  |
When it comes to corneal disease, you’d think no pain would be a promising sign. In some cases, the opposite may be true. Practitioners must know when to be concerned about corneal anesthesia.
The cornea is one of the most innervated tissues in the body, involving much more than simple sensation. Corneal nerves are vital to maintaining the integrity of the ocular surface, inducing reflex tearing and blinking and are a source of neurotrophic growth factors. Consequently, damage to corneal nerves can break down the corneal epithelium, impair wound healing and cause opportunistic infections, scarring, irregular astigmatism, stromal thinning leading to perforation and vision loss.
Damage to the ophthalmic branch of the trigeminal nerve can occur anywhere along the nerve pathway. Injury is commonly caused by surgery and herpetic infection but could also result from trauma, chemical burns, diabetes, corneal dystrophies and chronic use of contact lenses or medications.1-3 If there is no relevant history, think outside the box and order an MRI to rule out a compressive mass.1
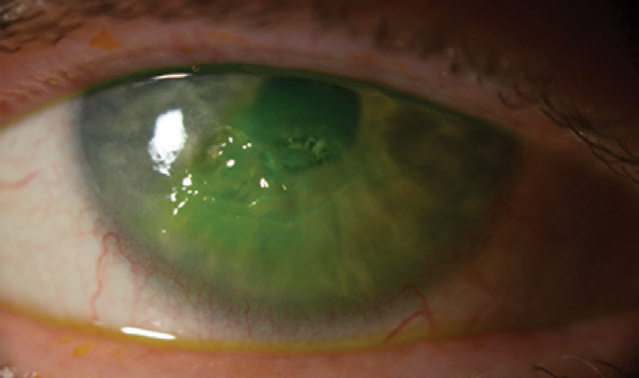 |
| The patient’s neurotrophic corneal ulcer after fluorescein instillation. Click image to enlarge. |
Sizing Up NK
Gather a comprehensive patient history and perform a thorough examination followed by a neurotrophic cornea work-up, which is as low-tech as using vital dyes and testing corneal sensitivity with the tip of dental floss or a wisp of cotton. Things become complicated when a patient develops neurotrophic keratitis (NK).
NK carries a high burden, taking its toll on ocular health and patient quality of life. Thankfully, this orphan disease is rare, affecting only five in 10,000 people, and many patients respond to ocular lubrication, bandage contact lenses, amniotic membranes and autologous serum.3 Once stable, NK patients can benefit from a scleral lens to constantly bathe the cornea.
However, supportive treatment may not always be enough. For a long time, the only other options included invasive surgery and Botox of the eyelid to induce ptosis. These options can limit vision, although a conjunctival flap is usually only considered in eyes that already have low potential, and significantly impact cosmesis.
Approach NK conservatively and use the most successful, least intrusive treatment. Thankfully, we now have tools for more recalcitrant cases. A study explored treating refractory neurotrophic corneal ulcers with topical insulin drops and found that every patient re-epithelialized in one to four weeks.4 Oxervate (cenegermin, Dompé) became the first FDA-approved treatment for NK in the summer of 2018 and is also the first to address the underlying cause of neurotropic disease by restoring corneal innervation. A pair of studies showed complete epithelial healing in 70% of patients after eight weeks of treatment and only 28% of controls.5
To acquire Oxervate, your office must fill out a form that your patient signs so that Accredo, the only pharmacy in the United States that fills the medication, can conduct an insurance benefits investigation. This form acts as both the prescription and the patient’s insurance information. Once received, Oxervate has to be refrigerated until opened and kept below 77°F after.5 Dosage is one drop six times a day instilled in two-hour intervals for an eight-week period. Your patient will need to be dexterous when opening the blister pack, adding the tip to the vial and using the pipette to draw and administer the medication.
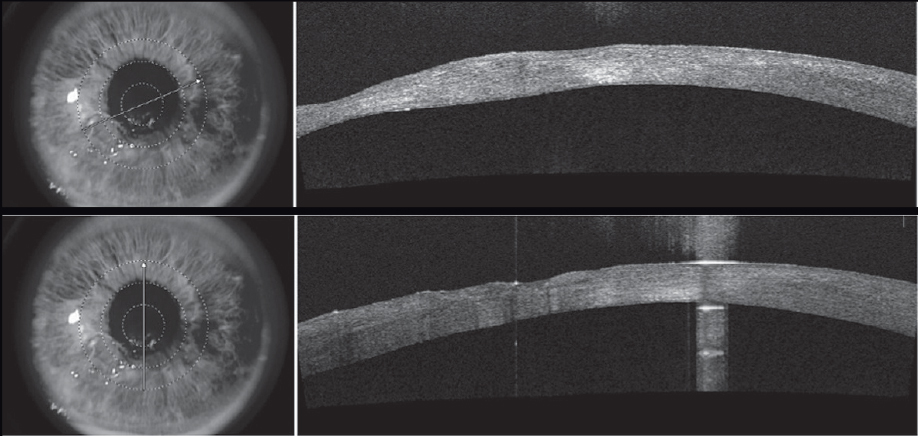 |
| Corneal OCT shows scarring, thinning and a highly irregular corneal surface due to the neurotrophic ulcer. Click image to enlarge. |
Case Study
A 76-year-old female presented with classic NK complaints—a comfortable but red right eye with worsening blurry vision. She had received successful treatment for herpes zoster ophthalmicus (HZO) 12 years earlier. Her shingles had resolved without any trigeminal neuralgia. She experienced no pain and could not feel the entrenched neurotrophic ulcer on her cornea.
The patient’s visual acuity was 20/200 OD with a large epithelial defect that showed mild thinning. She underwent epithelial debridement of dead tissue and started Vigamox (moxifloxacin, Novartis) QID, prophylactic famciclovir 250mg TID and preservative-free artificial tears Q1H. Her first Prokera (Bio-Tissue) was inserted at her next visit just days later.
Over the next year and a half, the patient was seen at least every three months, but often several times a month. Additional interventions included doxycycline when an ulcer began to thin, prednisolone in an attempt to limit scarring, topical ganciclovir when an active viral infection was suspected and a bland ointment for better lubrication at night. The latter was especially needed when she started developing filamentary keratitis. In an attempt to keep the cornea lubricated, a punctal plug was inserted. She was kept on an antiviral, antibiotic lubrication and an endless cycle of Prokeras. It was difficult to keep her epithelialized, even with amniotic membranes, so bandage contact lenses were not attempted.
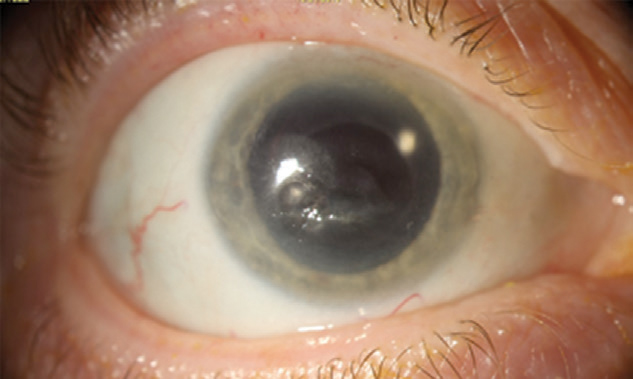 |
| A scarred but fully healed cornea following recurrent NK. Click image to enlarge. |
When the patient seemed stable enough to go a few months between appointments, she was asked to self-monitor for redness, vision changes and light sensitivity. Her cornea was scarred, and her vision was blurry, but the true danger lay in her inability to feel any recurrence.
A colleague had cared for the patient before he retired and her case became my responsibility. The release of Oxervate, a recombinant nerve growth factor, just months prior could not have come at a better time. I introduced the idea of this orphan drug to the patient who, despite learning that it was new and tricky to acquire, was excited about a fresh option when she had already exhausted so many.
The patient’s last Prokera was removed last March, and she started Oxervate in April. By June, she had finished the full course. Despite corneal neovascularization and scarring following her recurrent corneal disease, her epithelium was intact, and her cornea was the clearest it had been. For the first time, on a low prophylactic dose of famciclovir and with frequent lubrication, her cornea stabilized and suffered no erosions. At this point, we could finally start discussing options to improve her vision, such as a deep anterior lamellar keratoplasty (DALK) corneal transplant, cataract extraction or a combination procedure. The goal is always to perform surgery on a quiet eye to limit the risk of rejection or HZO reactivation.
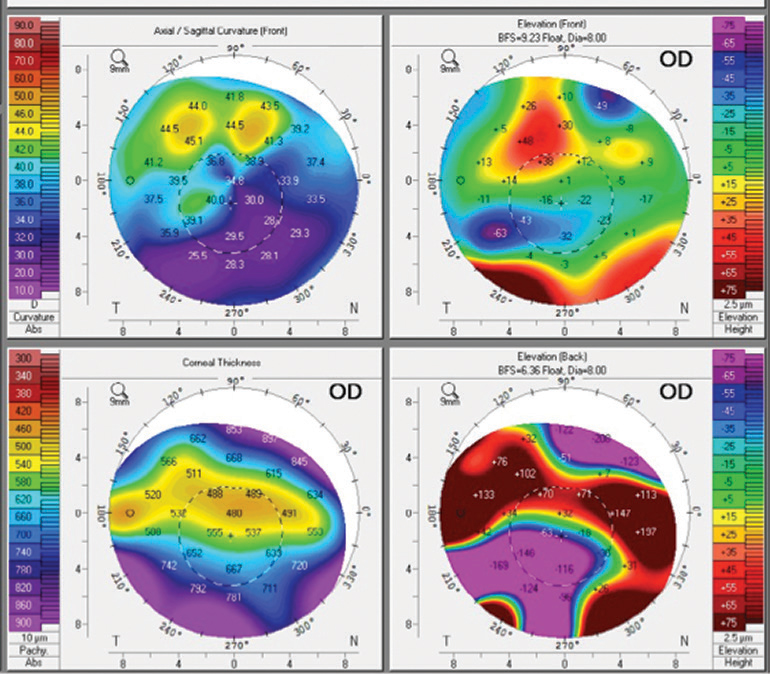 |
| Irregular corneal astigmatism with inferior flattening secondary to corneal scarring. Click image to enlarge. |
The patient deferred a DALK, which has a high risk of failure in NK eyes due to poor healing, but was cleared for cataract extraction in September. She understood that her vision would still be significantly limited secondary to her irregular corneal astigmatism and scarring and that hers was a higher-risk surgery. She started taking famciclovir 250mg TID one week prior to the procedure, which she continued for one month before returning to her previous prophylactic dose of QD.
The patient recently left the post-op period and is happy with her visual outcome of 20/200 OD without correction, pinholed to 20/50. She elected to pursue the nonsurgical route and wear scleral lenses. It has been about a year since the patient’s last NK event.
Corneal nerves play a vital role in ocular health, so when diminished corneal sensitivity threatens vision, it is our duty to educate patients on all available treatment options. Where we once had no medical intervention for NK, we now have a drug that gets at the root of the disease.
| 1. Versura P, Giannaccare G, Pellegrini M, et al. Neurotrophic keratitis: current challenges and future prospects. Eye and Brain. 2018;10:37-45. 2. Yang AY, Chow J, Liu J. Corneal innervation and sensation: the eye and beyond. Yale J Biol Med. 2018;91(1):13-21. 3. Sacchetti M, Lambiase A. Neurotrophic factors and corneal nerve regeneration. Neural Regen Res. 2017;12(8):1220-4. 4. Wang AL, Weinlander E, Metcalf BM, et al. Use of topical insulin to treat refractory neurotrophic corneal ulcers. Cornea. 2017;36(11):1426-8. 5. Oxervate prescribing information. U.S. Food and Drug Administration. www.accessdata.fda.gov/drugsatfda_docs/label/2018/761094s000lbl.pdf. Accessed December 30, 2019. |


