 New research into corneal staining associated with multipurpose solutions has sparked a name change and a new understanding among practitioners. Formerly known as solution-induced corneal staining (SICS), the new term—preservative-associated transient hyperfluorescence (PATH)—reflects that this is neither true corneal staining nor an indication of lens/solution bioincompatibility.
New research into corneal staining associated with multipurpose solutions has sparked a name change and a new understanding among practitioners. Formerly known as solution-induced corneal staining (SICS), the new term—preservative-associated transient hyperfluorescence (PATH)—reflects that this is neither true corneal staining nor an indication of lens/solution bioincompatibility.
What is PATH?
PATH is asymptomatic transient superficial corneal staining associated with the release of biocides from a contact lens. Importantly, it has not been correlated with pathology, infection or inflammation of the cornea. The staining has an onset between 30 minutes and four hours after lens insertion, and resolves within six to eight hours.1,2 In contrast, true corneal staining has a longer duration, varies in depth and fluorescein take up, and may have stromal streaming. It is also associated with other signs, such as hyperemia or infiltrates and future complications.
Contact lens solution preservatives—such as polyhexylmethyl biguanide (PHMB), Polyquad and Aldox—are absorbed into all soft contact lens materials, albeit in different amounts depending on the lens material, the specific preservative and the formulation of the care product.3,4 The various preservatives are then released from the lens at different rates. The Polyquad and Aldox release is greatest at 30 minutes, while PHMB is released between one to four hours after absorption.5,6 The release rates depend on the preservative and the lens material and coincides with the peak observable PATH.
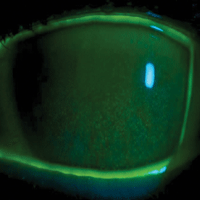
PATH staining.
Positively-charged molecules, such as the cationic preservatives in contact lens solutions, are attracted to the negatively-charged fluorescein (FL). Recent research suggests that the cationic lens solution biocides may associate with the anionic sites on the corneal epithelium and, concurrently with the anionic fluorescein, effectively bond the fluorescein to the corneal epithelial surface. The FL and preservative bonding strength varies between the different types of preservatives; FL with Polyquad is weaker than FL with PHMB.2
There may be predisposing factors that can increase one’s susceptibility to preservative-associated staining. One study found that 36% of contact lens wearers were “stainers.”7 Furthermore, there were no significant differences between strainers and non-stainers in regard to age, sex, refraction or keratometry. Stainers were more likely to show upper and lower palpebral hyperemia and tarsal roughness. The study did find that poor tear quality was related to preservative-associated staining.7
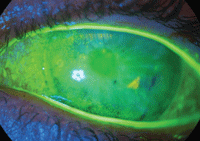
True corneal staining secondary to staph marginal keratitis.
The varying uptake and release patterns of the preservative in the lens/solution combinations, as well as the intrinsic strength of the preservative bond with either the cornea or FL, and the status of the tear quality, help explain the transient nature and high degree of variability of this phenomenon. It is important to differentiate PATH from the more clinically significant staining that results from pathological conditions so that appropriate intervention—monitoring, lens/solution changes or medical treatment—can be administered.
1. Garofalo RJ, Dassanayake N, Carey C, et al. Corneal staining and subjective symptoms with multipurpose solutions as a function of time. Eye Contact Lens. 2005 Jul;31(4):166-74.
2. Bright F, Merchea M, Kraut N, et al. A preservative-and-fluorescein interaction model for benign multipurpose solution associated corneal hyperfluorescence. Cornea. 2012 Dec;31(12);1480-8.
3. Bandamwar KL, Garrett Q, Cheung D, et al. Onset time course of solution induced corneal staining. Cont Lens Anterior Eye. 2010 Aug;33(4):199-201.
4. Fonn D, Peterson R, Woods C. Corneal staining as a response to contact lens wear. Eye Contact Lens. 2010 Sep;36(5): 318-21.
5. Powell CH, Lally JM, Hoong LD, Huth SW. Lipophillic versus hydrodynamic modes of uptake and release by contact lenses of active entities used in multipurpose solutions. Cont Lens Anterior Eye. 2010 Feb;33(1):9-18.
6. Dassanayake NL, Garofalo R, Carey R, et al. Correlating biocide uptake and release profiles with corneal staining and subjective symptoms. Invest Ophthalmol Vis Sci. 2005;46:e-abstract 915.
7. Young G, Canavan K, Jones S, Hunt C. Predisposing factors for solution-induced corneal staining. Optom Vis Sci. 2012 Nov;89(11):1582-9.


