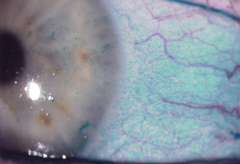
 |
| Sleeping on your back could help alleviate symptoms of dry eye. Image: Hank D. Perry, MD |
Back to Sleep for Dry Eye
A new study adds yet another facet to the already complicated conditions of dry eye disease (DED) and meibomian gland dysfunction (MGD): sleep position.
“Meibomian gland disease is often very asymmetric,” says Hank D. Perry, MD, of Ophthalmic Consultants of Long Island in Rockville Centre, NY, and the Department of Ophthalmology, Nassau University Medical Center in East Meadow, NY. “When you question these patients, you frequently find they usually sleep on the side with the more severe meibomian gland disease. So there is a relationship between the way they sleep and presenting problems of meibomian gland disease, and dry eye disease in general.”
To learn more about the possible association, Dr. Perry and colleagues asked 100 patients with DED symptoms and 25 controls to complete a sleep questionnaire and the Ocular Surface Disease Index (OSDI). While other studies have focused on nocturnal lagophthalmos, sleep apnea and floppy eyelid syndrome, this is the first to look at sleep position specifically, with more diagnostic parameters such as OSDI scoring, tear osmolarity, lissamine staining and Schirmer I testing, the authors said in the study.
In comparing the outcomes, they found a statistically significant difference with back sleeping compared to left side sleeping using lissamine green staining. They also found the OSDI scores were elevated in patients who slept on their sides compared with those who slept on their back. However, they found no statistically significant correlation between the sleep position and degree of MGD.
“We found that some patients got better just by changing their sleep patterns,” Dr. Perry says. “Not the easiest thing to do, but it can be done.” He also notes only 10% of the population sleeps on their backs, despite its benefits for those with MGD and dry eye.
“It’s another step [in dry eye care] that can help in terms of treating very difficult, complicated patients,” he says. “Something as simple as going from sleeping on their stomach to sleeping on their back can make a world of difference—and this is especially true for patients with severe meibomian gland disease, floppy eyelid syndrome” and other lid-involved conditions.
Eventually, Dr. Perry and his fellow researchers hope objective findings from a sleep center will further support the correlation uncovered by the patient questionnaires. Still, the current findings should lead many practitioners to add sleep patterns to their list of patient questions. “In some ways, it’s easier than having the patient take artificial tears or have them do lid hygiene,” Dr. Perry says. “It’s at least worth mentioning that their sleep pattern can affect the disease. It just makes sense.”
Alevi D, Perry HD, Wedel A, et al. Effect of sleep position on the ocular surface. Cornea. 2017;36:567-71.
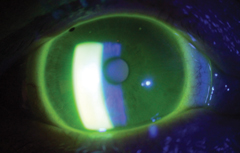 |
| A good scleral lens fit depends on the right diameter and thickness. Photo: Michael J. Lipson, OD |
The SCOPE of Scleral Lenses
After looking at current scleral lens prescription and management practices, a recent survey-based study found a considerable amount of consensus among participating practitioners.1 The Scleral Lenses in Current Ophthalmic Practice: an Evaluation (SCOPE) study surveyed 723 practitioners, all of whom had fit at least five patients with scleral lenses.
For instance, the survey showed 57% to 60% of respondents recommended using nonpreserved saline to fill the bowl of the lens prior to application. “To see such a large majority recommending preservative-free solutions for application of scleral lenses is a very strong recommendation,” says Michael J. Lipson, OD, clinical assistant professor at University of Michigan’s Kellogg Eye Center and vice president of the Scleral Lens Education Society. “Finding the right solution or ‘cocktail’ of solutions can be the difference between success and failure of a scleral lens fitting.”
Dr. Lipson sees this as a possible area for future scleral research and product development. “It’s a balance of cleansing, lubrication and overall biocompatibility that is required to make the patient comfortable and keep their vision and the cornea clear. We need to develop an application solution—sort of like ‘Gatorade for scleral lenses’—that provides natural electrolytes and maintains a clean, wet lens surface.”
The study also showed 73% of practitioners recommended mid-day lens removal on some, most or all days and 65% prescribed scleral lenses between 15mm and 17mm in diameter. According to Dr. Lipson, practitioners will find the most success when using various diameters to custom fit patients based on individual needs.
The authors believe more research is needed to develop evidence-based guidelines for scleral lens fitting; Dr. Lipson concurs. “The main issue is practitioner training and education,” he says. “Practitioners taking on sclerals for the first time should attend fitting workshops, webinars and educational meetings to know whom to fit, how to fit and what to look for during fitting and follow-up care to assure ideal visual and eye health results.”
Still, the SCOPE study could potentially serve as a starting point for practitioners new to sclerals. Also, the general agreement of those surveyed shows that practices are moving forward together in scleral fitting protocols. “As they are now, scleral lenses are generally very successful and can be life-changing for these challenging eyes,” says Dr. Lipson.
1. Harthan J, Nau CB, Barr J, et al. Scleral lens prescription and management practices: the SCOPE study. Eye & Contact Lens. 2017;0:1–5.
| In Brief |
| After following 500 patients post Descemet membrane endothelial keratoplasty (DMEK), researchers found outcomes remain excellent up to two years after the procedure. The study evaluated best-corrected visual acuity (BCVA), central endothelial cell density and central corneal thickness before and at six, 12 and 24 months after DMEK. At one year post-procedure, 81% of eyes recorded a BCVA of ≥20/25, 49% reached ≥20/20 and 15% achieved ≥20/18—and remained stable up to 24 months. Retransplantation was required in only 6.4% of eyes, and few participants had long-term complications such as secondary graft failure (1.4%) and allograft rejection (1.4%). Peraza-Nieves J, Baydoun L, Dapena I, et al. Two-year clinical outcome of 500 consecutive cases undergoing Descemet membrane endothelial keratoplasty. Cornea. April 12, 2017. [epub ahead of print]. When studying tear neuropeptide levels in 20 contact lens wearers and 20 non-wearers, researchers uncovered a relationship between nerve density, calcitonin gene-related peptide concentration and corneal sensitivity. Lens wearers had higher Ocular Comfort Index scores and tear osmolarity compared with the non-wearers, but lower noninvasive tear break-up times. Tear neuropeptide concentrations, nerve morphology and ocular surface sensitivity were no different between the two groups. The authors conclude that contact lens wear does not seem to alter markers of corneal neurobiology and sensory function, despite worse tear function. Golebiowski B, Chao C, Stapleton F, Jalbert I. Corneal nerve morphology, sensitivity, and tear neuropeptides in contact lens wear. Optom Vis Sci. 2017;94(4):534–42. Researchers who recently looked into the in vitro extended drug reservoir function of human amniotic membranes (HAM) were able to achieve sustained release of moxifloxacin from HAM for up to seven weeks. The study, which involved a process of soaking HAM buttons of different thicknesses with moxifloxacin and closely assessing the release kinetics, may have revealed a new application for sustained drug delivery through a biological bandage system when dealing with bacterial keratitis. Results also showed significantly higher entrapment efficiency for moxifloxacin in thicker HAM compared with thinner. Yelchuri ML, Bhagyashree M, Nilam G, et al. In vitro evaluation of the drug reservoir function of human amniotic membrane using moxifloxacin as a model drug. Cornea. 2017; 36(5):594–9. |


