Nowadays, practitioners are playing a more active role in comanaging patients after ocular surgery. This involves optimizing refractive results and, less frequently, restoring visual acuity to irregular ocular surfaces that may have been altered by infection or trauma (including the surgery itself). Specialty contact lenses are one solution, offering numerous possibilities that make it easier than ever to achieve efficient, accurate results. This article summarizes the steps required to determine the right lens for each post-op patient. In most circumstances, customization is key.
Step 1: Know What You Are Working With
First and foremost, we must under-stand what kind of ocular surface we are dealing with. This starts with good communication with the surgeon about the surgery, expected visual outcomes, post-op medications, possible complications and long-term issues. Learning the endothelial cell count after a corneal graft will dictate what lenses we can consider. If the patient had epithelial healing issues post-op, the contact lens fit will likely necessitate piggybacking or scleral lenses to vault over the fragile surface (Figure 1).
 |
| Fig. 1. Scleral lens over corneal rings. Click image to enlarge. |
If this information is not readily available, obtain a thorough case history from the patient during their first follow-up. Use this time to take the patient’s expectations and feelings toward contact lens wear into consideration. For some patients, especially if they aren’t interested or able to be fit in specialty contact lenses, glasses are the modality of choice and offer decent vision. Keeping these elements in mind, determine the best strategy and put together a personalized plan with which to move forward.
Step 2: Take a Refraction
Although this may seem obvious, refraction should be the next step in addressing the patient’s ocular needs after surgery.
Electronic refraction may be misleading in some cases of corneal irregularity and severe eye dryness. This is why I prefer starting my refractions with the tried-and-true retinoscopy technique. Not only can objective refraction be evaluated quickly, but it also provides a lot of useful information. For example, the presence of ocular media opacities may potentially limit visual outcomes. The quality of the red reflex says a lot about higher-order aberrations and surface irregularities. What you see through retroillumination is perceived by the patient and may disturb their vision. If you can’t see well through this medium, this will also hold true for the patient. In the past, topography was frequently dictated by retinoscopy findings, which later showed forme fruste keratoconus on the posterior corneal surface. This type of abnormality might not have been detected if not for a quick but efficient retinoscopy.
Retinoscopy is even more important when contact lenses are introduced, as it can easily highlight over-correction and residual astigmatism. The best example is the patient between 36 and 40 years old who complains about vision but sees 20/20 perfectly. Most of the time, retinoscopy will show masked astigmatism and hyperopia. Knowing that binocular vision imbalance may mimic dry eye signs and symptoms, adjust the power and try toric lenses to reduce discomfort by enhancing the effect of accommodation and convergence.1
Subjective refraction, on the other hand, determines the potential visual acuity and may dictate the limits of contact lens selection in cases of powers or axes outside the available parameters. Whenever 20/20 is not achieved during subjective refraction, you can obtain pinhole acuity to evaluate visual potential.
Step 3: Optimize the Ocular Surface
For post-surgery patients, it is easier to select specialty contact lenses if you can back your decision with a series of clinical tests that include aberrometry, corneal and conjunctival topography and OCT imaging. Before assessing the ocular surface, make sure to optimize it, as ocular dryness, low tear break-up time and an unstable tear film can negatively influence results. You will have false negatives or positives, incorrect values and misleading information if the surface of the eye is not uniform enough to allow light rays to travel without disturbance or accurately reflect the ocular surface. Even a refraction will not be as precise in the presence of moderate-to-severe eye dryness.
According to the DEWS II report, a dry eye assessment should start with a few questions about patient symptoms and artificial tear necessity.2 Using artificial tears more than two or three times a day indicates chronic eye dryness. Testing is then required to screen the face and lids for rosacea and to analyze tear break-up time, corneal staining and meibomian gland expression of the nasal quadrant of the inferior lid.
Significant findings should motivate practitioners to conduct a more comprehensive dry eye workup that will help assess and treat the condition to limit adverse events and optimize refractive outcomes. Even with normal findings, it is still considered adequate to instill one to two drops of artificial tears before assessing the ocular surface and performing ocular topography.
Step 4: Map the Cornea
The logical next step is to objectively log the patient’s eye condition by mapping the cornea and the conjunctiva to get the most data possible.
Corneal topography captures and displays the irregularity of the surface after surgery. For post-op patients, an axial map is useful when viewing the cornea and establishing the presence of regular or irregular astigmatism. An elevation map is also essential because it highlights the highest point of the cornea you need to consider when fitting specialty lenses—and scleral lenses in particular—and pinpoints relative differences in quadrant elevation.
One study found that patients with 350µm or less of corneal elevation difference along the greatest meridian of change have an 88.2% chance of success with a corneal gas permeable lens vs. a scleral lens.3 This map also provides practitioners with the best fit sphere, which represents the most valuable starting point when fitting gas permeable lenses, as the goal is to align the base curve of the lens with the best fit sphere value. Mapping the conjunctival surface also aids in scleral lens fittings. In fact, this is the only objective method to determine how much toricity is required (peripheral haptics), and it helps predict lens rotation (along the steepest conjunctival meridian) in cases of front-toric lens designs. Remember that 65% of the sclera is highly irregular, asymmetric and non-rotational and has various amounts of toricity.4
When visual acuity is not as sharp as expected or patients complain about shadowing along images, aberrometry becomes an essential step toward understanding and improving the patient’s condition. Conducting aberrometry when a patient is wearing their lenses is useful in diagnosing the presence of higher-order aberrations, especially coma, which is often mistaken for residual or induced astigmatism in scleral lens wear. In my experience, this is particularly true for patients with irregular corneas, whom I am able to refract to at least 20/30 in glasses. In such cases, hybrid lenses may be a better option.
Anterior segment OCT allows practitioners to evaluate the cornea, the conjunctiva, the corneo-scleral junction (the most important factor driving soft lens behavior) and the surface-to-lens relationship.5 In cases of scleral lenses specifically, measurements include lens conjunctival compression, position, thickness and tear reservoir thickness. With proper and complete imaging of the anterior surface, it is possible to execute specialty contact lens fittings effectively.
Step 5: Select the Right Lens
Now it’s time for the fitting process. A flowchart can help you determine the go-to lenses for each irregular cornea you encounter (Figure 2).
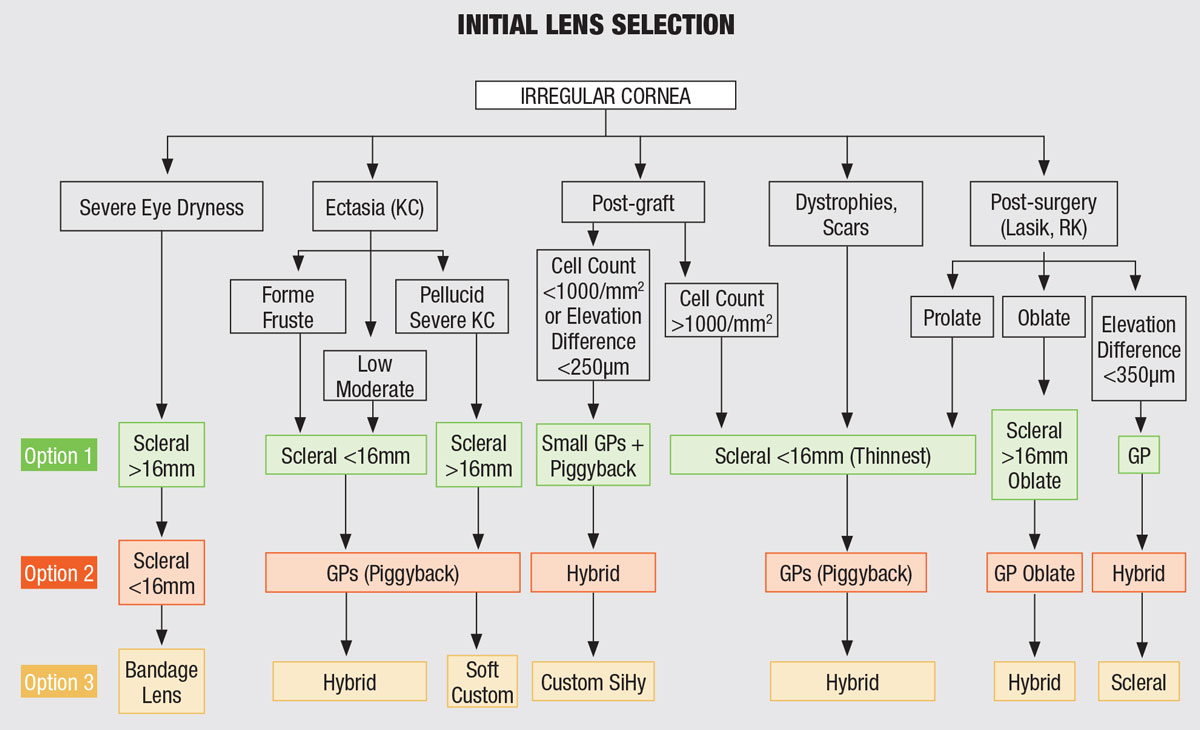 |
| Fig. 2. This flowchart may come in handy when deciding on a lens for an irregular cornea. Click image to enlarge. |
I gravitate toward scleral lenses under most circumstances. Marginal eye dryness can be better addressed with scleral lenses, particularly in an aging population. However, smaller gas permeable lenses may be justified in certain circumstances, such as: whenever corneal health is at risk (e.g., when a patient has a low endothelial cell count), the corneal surface elevation difference is less than 350µm, the scleral profile is highly irregular or scleral lens designs cannot be fully customized.
Hybrid lenses are still a valuable option, but they cannot be designed as front-toric lenses when residual astigmatism is in play. The fact that the gas permeable carrier junction lies on the cornea could also limit their use. The absence of a tear reservoir may represent another shortcoming in the presence of significant eye dryness.
Presbyopia must remain at the forefront of practitioners’ minds, as many patients have declined or were not offered this type of multifocal implant. If astigmatism and presbyopia are present, gas permeable lenses, hybrids and sclerals are all choices that can compensate for these combined refractive errors.
Contact lenses don’t have to be the only option for presbyopes struggling with fi t, comfort or both. Wearing contacts for specific activities and glasses the rest of the time is a good way to overcome their concerns while still correcting their vision. Daily disposable lenses are the number one option for ocular health and convenience when part-time wear is involved.
I like to relate the diverse nature of vision, especially for active presbyopes, to shoes. When patients realize they have different shoes for different activities, it becomes easier for them to understand that vision operates in a similar way. There is no logical reason to shy away from offering presbyopic correction to post-op patients if the need to see clearly at near has not already been addressed with multifocal implants.
When sclerals, gas permeable lenses and hybrids are not well tolerated, you can consider soft custom lenses. However, these lenses are thicker and have low oxygen permeability. They should be fitted loosely to favor tear exchange, which is the only way to supply oxygen to the cornea when they are worn.
When dealing with post-op patients with normal prolate corneas, as is often the case after cataract surgery, silicone hydrogel lenses are a preferred option, especially if the patient has worn this type of modality in the past. Any residual refractive error can be easily corrected with disposable lenses.
Following each modality’s fitting guide provided by the manufacturer is a must. Doing so could yield success rates as high as 90% with the first or second pair tried.6 Satisfied patients are your biggest advocates outside of the office.
Step 6: Determine a Care Regimen
Chemical exposure secondary to preservatives and buffer agents may generate chronic reactions if the proper precautions aren’t taken. The presence of these ingredients may also cause cross-reactions with other topical products (artificial tears, glaucoma medications, steroids, etc.).
Hydrogen peroxide systems are typically recommended because they do not contain preservative agents and are compatible with most of the lenses on the market. For sensitive patients, keep things simple and alleviate formulation with wetting agents. The same holds true for comfort drops, artificial tears and topical medications, which should be prescribed non-preserved whenever possible.
Topical medications do not necessarily represent a contraindication for contact lens wear if the dosage is equal to or less than BID. Otherwise, it may be convenient for the patient to remove their lenses and apply the drug during the day.
Step 7: Schedule Regular Follow-ups
Post-surgery ocular health changes over time as a patient ages, and today’s positive outcome could become tomorrow’s terrible nightmare—think radial keratotomy patients who tend to be happy in the months following the procedure but then develop fluctuating vision and severe refractive shifts toward hyperopia over time. Regular follow-ups are needed to continue to optimize visual correction outcomes, especially when contact lenses are in use, and improve compliance. For new contact lens wearers, the initial three months post-fitting are the most important, as one study demonstrated that 25% of dropouts occur within the first 90 days of wear.7 It goes without saying that the presence of comorbidities, such as diabetes and glaucoma, should dictate a specific and personalized follow-up schedule.
Post-surgery patients can be challenging in many ways. Luckily, numerous contact lens options exist to meet their visual and ocular health needs. Doing your best to customize a plan for each of your patients is all the more rewarding when you achieve successful outcomes.
Dr. Michaud is chief of the contact lens department at the University of Montreal, where he teaches and practices. He is a Fellow of the American Academy of Optometry (Diplomate), the British Contact Lens Association, the Scleral Lens Education Society and the European Academy of Optometry. Dr. Michaud has published numerous articles in peer-reviewed journals and publications, is the author of more than 35 book chapters and regularly speaks at major optometric and ophthalmology meetings around the world.
| 1. Rueff EM, King-Smith PE, Bailey MD. Can binocular vision disorders contribute to contact lens discomfort? Optom Vis Sci. 2015;92(9):e214-21. 2. Craig JP, Nelson JD, Azar DT, et al. TFOS DEWS II report executive summary. Ocul Surf. 2017;15(4):802-12. 3. Zheng F, Caroline P, Kojima R, et al. Corneal elevation differences and the initial selection of corneal and scleral contact lens. Poster presented at the Global Specialty Lens Symposium, January 22-25, 2015; Las Vegas, NV. 4. DeNaeyer G, Sanders D, van der Worp E, et al. Qualitative assessment of scleral shape patterns using a new wide field ocular surface elevation topographer. J Cont Lens Resear Sci. 2017;1(1):12-22. 5. Young G. Ocular sagittal height and soft contact lens fit. J Br Cont Lens Ass. 1992;15(1):45-9. 6. Brujic M, Kading D. Multifocals: A roadmap to success. Review of Cornea and Contact Lenses. www.reviewofcon-tactlenses.com/article/multifocals-a-road-map-to-success. September 15, 2018. Accessed December 19, 2019. 7. Sulley A, Young G, Hunt C. Factors in the success of new contact lens wearers. Cont Lens Ant Eye. 2017;40(1):15-24. |


