Dry eye syndrome is a common condition found in most optometric eye care practices, and can often make the difference between success and failure in a contact lens patient. It can affect patients in both soft and gas-permeable lenses. Recent studies have found that 52.7% of contact lens wearers suffer from dry eye––a much higher percentage than found in the general population, which is estimated to range between 14% and 33%.1
Dry Eyes in the CL Wearer
 Dry eye disease yields symptomatic and objective findings that are all related to an insufficiency of the tear film––whether caused by an overall decrease in tear volume or an increase in tear evaporation.2 Some common patient complaints associated with dry eye may include burning, grittiness, foreign body sensation or blurred vision.
Dry eye disease yields symptomatic and objective findings that are all related to an insufficiency of the tear film––whether caused by an overall decrease in tear volume or an increase in tear evaporation.2 Some common patient complaints associated with dry eye may include burning, grittiness, foreign body sensation or blurred vision.
For a patient to be successful with contact lens wear, he or she needs to have a stable tear film. The tear film is what keeps the lenses hydrated, ensures adequate oxygen transmission, and reduces the chances of bacterial contamination of the contact lenses. Contact lens wear can reduce corneal sensitivity and alter the normal tear secretion needed to maintain a healthy ocular surface.3
Patients find the easiest and greatest relief of contact lens-induced dryness following lens removal. Unresolved dryness issues can cause a patient to discontinue contact lens wear permanently,4,5 which can lead to an increase in contact lens wearer drop out rates.5
The use of artificial tears can increase a patient’s comfort, performance and contact lens tolerance. A stable tear film is ideal for a successful contact lens fit.3 A combination of tears that works on unique portions of the tear film may be ideal for optimal therapy.6 Studies have shown that more than 50% of contact lens wearers have tried artificial tears to relieve their discomfort; yet, more than 50% of that population reported no relief.1
In an ideal situation, the tear film should surround a contact lens on both sides.7 If tears evaporate or break up too quickly, they are not able to constantly surround a contact lens, leading to discomfort. It has been shown that tear film break-up time, volume and stability are all lower in intolerant contact lens wearers.7 Studies have also shown that a decrease in contrast sensitivity and visual acuity in contact lens wearers can be attributed to a change in their tear layer. Low-viscosity artificial tears helped improve the patient’s visual acuity, while artificial tears with higher viscosity did not improve visual performance.8
The tear film is comprised of three distinct layers, each with its own purpose. The lipid layer is the most anterior layer and is made up mostly of meibomian oil and helps maintain the tear film stability, decrease evaporation and thicken the aqueous layer in order to create a smooth ocular surface. The middle layer––known as the aqueous layer––is produced by the lacrimal gland and makes up the majority of the tear film. It chiefly consists of water and proteins. The most posterior layer, the mucous layer, is made up of mucins and coats the cornea, attaching to the aqueous layer to create an even, stable tear film.6 Without this layer, the tears would not be anchored to the cornea.
Tear film deficiencies can be found at any of these layers and can cause symptoms of dryness with or without contact lens wear. Any of these tear film problems can occur simultaneously or independently.
 OTC Options for Contact Lens-Associated Dry Eye
OTC Options for Contact Lens-Associated Dry Eye
Over-the-counter (OTC) agents can be used to help patients manage their contact lens-related dry eye and prevent further complications.9 There are both on- and off-label options for these patients to increase their comfort and chances for successful contact lens wear. It is important to remember that preservatives are needed in all ophthalmic drops to prevent bacterial growth. Unfortunately, these preservatives can have a cytotoxic side effect to the eye with long-term use.10 For patients using drops as part of a frequent regimen, preservative-free drops or drops with fewer toxic preservatives may be a better option.
Not all artificial tears are created equally or are intended to affect the same layer of the tear film. It is very important that practitioners determine which layer of the tear film is causing the dry eye symptoms and contact lens discomfort before prescribing an artificial tear. A strong case history is also crucial, because symptoms may not always match objective findings.11 By using the correct artificial tear with contact lens wear, you can increase successful treatment and continuation of contact lens wear.
Studies have shown that tears that merely increase tear volume can cause symptoms to worsen in patients with a lipid deficiency.6 Advances in research and production have created several options with unique mechanisms of action and formulation. Recent reports have focused on decreased lipid layer thickness in patients with dry eye symptoms.12 Unfortunately, there are no commercially available systems on the market to measure a patient’s lipid thickness for objective dry eye findings. Manufacturers are also taking a newer approach of using artificial tears for osmoprotection.9
 Review of Available Agents
Review of Available Agents
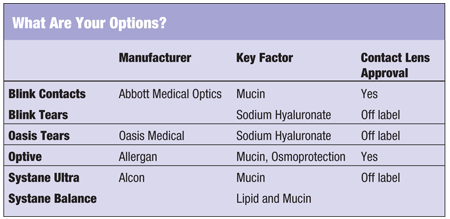
• Systane Balance (Alcon) was developed to help with meibomian gland dysfunction (MGD). Patients with MGD experience changes in the lipid layer of their tears. Systane Balance is the only artificial tear on the market that is able to work on the lipid and mucin layer simultaneously. It has two mechanisms of action to increase comfort and decrease dryness. Like Soothe XP, it is a lipid emulsion drop that permits restoration of the lipid layer (LipiTech System), thereby thickening the aqueous layer. By thickening the lipid layer, evaporation decreases.
Systane Balance also contains the unique hydroxypropyl guar (HP-guar) system that strengthens the attachment of the aqueous layer to the glycocalyx-mucin interface. This further allows the aqueous layer to increase.13 Systane Balance has not been approved for use with contact lenses, but has been used off-label by many practitioners without any issues. Sorbitan tristearate is used to preserve Systane Balance drops, which are currently unavailable in a preservative-free option.
Systane and Systane Ultra also contain the guar system to increase the mucin aqueous interface, but do not contain a system to increase the lipid layer of the tears. Systane Ultra will cause less blur than the original Systane due its lower viscosity upon instillation, which is made possible by the addition of sorbitol to the formulation.13 Both Systane and Systane Ultra are available in preservative-free forms.
• Oasis Tear products (Oasis Medical) are available in two formulations: Oasis Tears and Oasis Tears Plus. The active ingredient in both formulations is glycerin. Oasis Tears also contain sodium hyaluronate, which has been shown to improve subjective and objective findings of dry eye. This is a naturally occurring polymer that is seen at ocular damage sites. It is believed to have anti-inflammatory properties as well. Many patients with dry eye symptoms will experience changes in their corneal epithelium that can cause erosions.15
Some studies have shown that sodium hyaluronate is actually able to help protect and promote healing of a dry eye patient’s corneal epithelium.15 Sodium hyaluronate has the ability to retain water, allowing for increased surface wettability. It can change viscosity upon blinking and is more viscous while the eye is open, which improves tear film break-up time and helps spread the agent more evenly across the ocular surface.16
Oasis Tears are most commonly prescribed in a non-preserved form, but have recently been released in a bottled preserved formulation. None of the Oasis Tear formulations have been approved for contact lens use. Off-label use of these products for contact lens wearers has been highly successful in our practice. Application before, during and after contact lens wear has allowed many of our patients to significantly increase their comfortable contact lens wearing time. Oasis Tears can be purchased only directly at a doctor’s office or online from Oasis Medical.
• Blink Tears (Abbott Medical Optics) Lubricating Eye Drops are designed to mimic the mucin layer of the tear film, and are able to bind to 1,000x their value of water. By improving the mucin layer of the tears, the tears become more stable and adhere better to the ocular surface. The improved adhesion increases the aqueous volume. These drops also contain sodium hyaluronate, but at a lower percentage and molecular weight than Oasis Tears.
The preservative stabilized oxychloro complex (SOC), found in all versions of Blink Tears, causes less damage to the ocular surface because it can convert into components that are naturally found in the eye. This decreases the chances of cytotoxic effects from continued use of artificial tears for dry eye symptoms.10 A recent study found that instilling a drop of Blink Contacts on the lens before insertion decreased objective and symptomatic findings of dry eye.17 Blink Contacts are approved for use with contact lenses, but Blink Tears may be used in an off-label application.
• Optive Eye Drops (Allergan) are designed to provide increased osmoprotection. Compromised tear films create a hypertonic state, while a healthy eye’s tear film is isotonic. A hypertonic tear film will cause osmotic stress because more water will leave the epithelial cells in an attempt to balance the osmotic state. As more tears leave the epithelial cells, the cornea becomes dehydrated and compromised, increasing dry eye symptoms.
Optive promotes epithelial cell stability under dry eye-induced hypotonic stress. These drops contain the active ingredient sodium carboxymethylcellulose (which acts as a lubricant) and glycerol (which acts as a moisturizer). The glycerol helps water bind to the cells. This combination aims to create osmoprotection by binding to the corneal cell surface to reduce water loss.18 Optive utilizes Purite as its preservative. Purite, which is similar to the SOC found in Blink Tears, disappears immediately as it converts into common tear components once it interacts with the ocular surface.10 Due to this mechanism, there is less concern about cytotoxic results from long-term use. Optive Eye Drops have been approved for use with contact lenses and is also available in preservative-free vials.
Identifying dry eye patients before and during contact lens wear is beneficial to both the patient and practitioner. By recognizing these patients and treating them accurately, we as practitioners can improve their contact lens comfort and chances of successful contact lens wear, as well as minimize contact lens drop out rates. By understanding the differences among OTC choices, we can educate our patients about the ideal choice for them. Currently, there is no cure for dry eye conditions, so we must do our best to control the symptoms and improve our patients’ experiences with contact lens wear.
1. Scaffidi RC, Korb DR. Comparison of the efficacy of two lipid emulsion eye drops in increasing tear film lipid layer thickness. Eye Contact Lens. 2007;33:38-44.
2. Lemp MA. Report of the National Eye Institute/Industry workshop on Clinical Trials in Dry Eyes. CLAO J 1995;21:221-32.
3. Foulks G. What is dry eye and what does it mean to the contact lens wearer? Eye & Contact Lens. 2003:29;96-100.
4. Chalmers RL, Begley OG. Dryness symptoms among an unselected clinical population with and without contact lens wear. Contact Lens & Anterior Eye. 2006;29:25-30.
5. Young G, Veys J, Pritchard N, Coleman S. A multi-centre study of lapsed contact lens wearers. Ophthalmology Physiology. 2002:22;516-527.
6. Calvao-Santos G, Borges C, Nunes S, et al. Efficacy of 3 difference artificial tears for the treatment of dry eye in frequent computer users and/or contact lens users. Eur J Ophthalmol. 2011;21(5):538-544.
7. Glasson MJ, Stapelton F, Kaey L, Willcox MDP. The effect of short term contact lens wear on the tear film and ocular surface characteristic of tolerant and intolerant wearers. Contact & Anterior Eye. 2006:29;41-47.
8. Ridder WH, Tomlinson A, Paugh J. Effect of preservatives in artificial tear solutions on tear film evaporation. Ophthalmology Physiology. 1991;2:48-52.
9. Kaercher T, Buchholz P, Kimmich F. Treatment of patients with keratoconjunctivitis sicca with Optive: results of a multicenter, open-label observational study Germany. Clin Ophthalmol. 2009:3;33-39.
10. Noecker R. Effects of common ophthalmic preservatives on ocular health. Adv Ther. 2001;18(5):205-215.
11. Blackie CA, Solomon JD, Scaffidi RC, et al. The relationship between dry eye symptoms and lipid layer thickness. Cornea. 2009:28(7);789-794.
12. Korb DR. The effect of two novel lubricant eye drops on tear film lipid layer thickness in subjects with dry eye symptoms. Optom Vis Sci. 2005;82:594-601.
13. Alcon PowerPoint presentation on Systane Ultra and Systane Balance products October 2011.
14. Cristensen MT, Blackie CA, Korb DR, et al. An evaluation of the performance of a novel lubricant eye drop. Poster presented at ARVO meeting 2010.
15. Aragona P, Papa V, Micali A, M. et al. Long term treatment with sodium hyaluronate-containing artificial tears reduces ocular surface damage in patients with dry eye. Br J Ophthalmol. 2002;86:181-184.
16. Rah M. Hyaluronan: A review of its properties, ophthalmic uses and research. Contact Lens Spectrum. April:2010.
17. Frenetter B, Michaud L. Evaluation of sodium hyaluronate lubricating drops used before insertion of contact lenses on symtomatology, severity and intensity of ocular dryness. University of Montreal, Poster presented at AAO Meeting 2011.
18. Kaercher T, Buchhilz P, Kimmich F. Treatment of patients with keratoconjunctivities sicca with Optive: results of a multicenter: open-label observation study in Germany. Clin Ophthalmol. 2009:3:33-39.
Dr. Brafman is Co-Director of Contact Lens Specialty Services at North Suburban Vision Consultants, Ltd. and an assistant clinical professor at the Illinois College of Optometry, Illinois Eye Institute.
Dr. Eiden is the President and Medical Director of North Suburban Vision Consultants, Ltd., and is an assistant clinical professor at the University of Illinois, Chicago, Department of Ophthalmology, Cornea and Contact Lens Service.


