 |  |
Although the gas-permeable (GP) lens market has been stable for the past several years, there is no denying the general decline in GP lens fitting since the introduction of soft contact lenses.1,2 Compared with the soft lens fitting experience, a survey found eye care practitioners often cite initial discomfort and increased chair time as reasons for hesitation with fitting GPs.3 Despite these perceived drawbacks, GP lenses are still highly regarded for their ocular health profile.
Avoiding the Limbus
Anatomically, the limbus is the junction between corneal and conjunctival tissue. In addition to containing a vascular supply and aqueous humor pathways, the limbus is also home to limbal epithelial stem cells (LESCs).4 These supply basal cells that migrate and proliferate into corneal epithelial cells. Mechanical disruption or hypoxia of these LESCs can lead to limbal hyperemia and corneal neovascularization.5 In the event of significant insult or injury, limbal stem cell deficiency (LSCD) can also occur. Although this condition is rare, the resultant epitheliopathy, corneal conjunctivalization and scarring can be devastating.6,7
While soft lenses drape over the entire cornea, small-diameter corneal GPs have minimal interaction with the limbus and typically avoid compression entirely. A review of the literature reveals only one case of LSCD attributed to GP lenses, while the remaining cases involve soft lenses.8 The incidence of corneal neovascularization is also lower with GP lenses than soft lenses, which may be attributed in part to the lack of limbal disruption.9
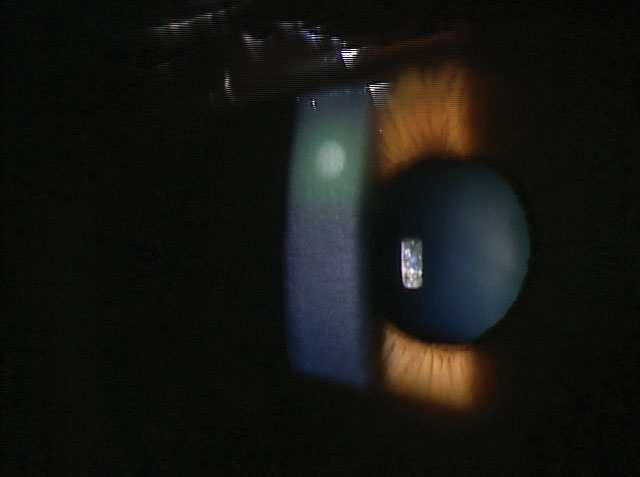 |
| Extended wear of SiHy lenses increases risk of symptomatic CIEs, such as a sterile marginal ulcer. |
The Role of Oxygen
In an open-eye environment, the atmosphere is the main supply of oxygen to the cornea. Placing a contact lens on the eye can reduce this supply by 8% to 15% depending on the oxygen permeability (Dk) of the lens.9 If oxygen levels decrease enough to leave the cornea in a hypoxic state, multiple physiological changes can occur, including epithelial defects, neovascularization, acute stromal edema and endothelial polymegethism.9
Commonly associated with hypoxia, corneal inflammatory events (CIEs) include infiltrative keratitis, sterile marginal ulcers and contact lens-associated red eye (CLARE). In the non-contact lens-wearing population, the prevalence of asymptomatic CIEs can be as high as 4%.10 Although incidence rates vary among studies, the relative risk of symptomatic CIEs increases significantly with extended contact lens wear for both higher Dk silicone hydrogel (SiHy) lenses and hydrogel lenses.11-13 One study compared the clinical success of 30-day continuous wear high Dk GP lenses with extended wear SiHy lenses. The GP group contained half as many adverse events as the SiHy group, with the study classifying two events as a CIE for the GP group and nine events for the SiHy group.14 Other studies have also confirmed lower rates of CIE for GP lenses.12,15
Even with similar Dk between GP and SiHy lenses, some speculate GPs are safer due to tear exchange. Compared with the general tear stasis underneath a soft lens, the smaller diameter of a GP lens promotes a flushing mechanism that improves oxygen tension reaching the corneal surface.16 Additionally, research shows tear film has bacteriostatic activity, although the exact mechanism is unknown.17
Reduced Microbial Load
When comparing SiHy and hydrogel lenses, SiHy lenses actually have higher risk of CIEs—suggesting hypoxia is not the only risk factor.18 While CIEs are considered sterile, many think bacterial bioburden plays a part to their pathogenesis. However, the larger concern of bacterial exposure is microbial keratitis (MK), which, although rare, can be sight threatening.
In one study, the annual incidence rate of MK among all contact lens wearers was 4.2 per 10,000.19 When comparing modalities, the rate for soft lens wearers was 1.9 per 10,000 and 11.9 for SiHys.19 When worn as extended wear, these rates increased to 19.5 for soft lenses and 25.4 for SiHys.19 However, in GP daily wearers the rate was only 1.2 per 10,000.19 An earlier study had similar annual incidence rates of 2.0 per 10,000 for GP lenses and 2.2 to 4.1 per 10,000 for daily soft lenses.20
All of this boils down to the fact that GP lenses have a lower risk profile for MK than soft lenses. In addition to having a 10 to 20 times greater tear exchange than a soft lens, GP lens surfaces have lower adherence rates of bacteria, according to at least one study.21,22
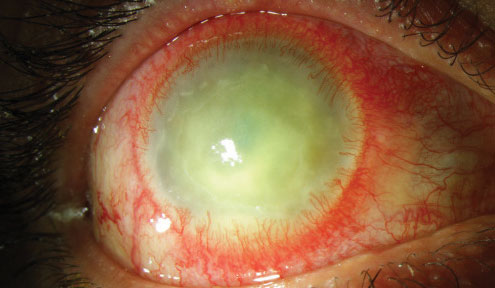 |
| Acanthamoeba keratitis seldom occurs in GP wearers. |
Another major concern for contact lens wearers is the exposure to Acanthamoeba present in most forms of water, including tap water. GP lens wearers are far more likely to rinse and store their lenses with tap water than soft lens wearers.23 Although the literature investigating Acanthamoeba keratitis and GP lenses have typically involved orthokeratology lenses, there are still relatively few reports of Acanthamoeba keratitis among GP wearers.24 The lack of water content in GP lenses, compared with soft lenses, is one contributing factor that significantly reduces the rate of Acanthamoeba’s adherence.25
Like any contact lens, GP lenses are not without risk or complication; however, when weighing the benefits of different lens modalities, practitioners should strongly consider GP lenses for their safety profile.
1. Nichols JJ. Contact lenses 2017. CL Spectrum. 2018;(1):20-25, 42. 2. Efron N, Nichols JJ, Woods CA, Morgan PB. Trends in US contact lens prescribing 2002 to 2014. Optom Vis Sci. 2015;92:758-67. 3. Gill FR, Murphy PJ, Purslow C. A survey of UK practitioner attitudes to the fitting of rigid gas permeable lenses. Ophthal Physiol Opt. 2010;30:731-9. 4. Van Busbirk EM. The anatomy of the limbus. Eye. 1989;3:101-8. 5. Papas EB. The role of hypoxia in the limbal vascular response to soft contact lens wear. Eye Contact Lens. 2003;29(1 Suppl):S72-4; discussion S83-4, S192-194. 6. Sangwan VS. Limbal stem cells in health and disease. Biosci Rep. 2001;21(4):385-405. 7. Chan CC, Holland EJ. Severe limbal stem cell deficiency from contact lens wear: patient clinical features. Am J Ophthalmol. 2013;155(3):544-9. 8. Rossen J, Amram A, Milani B, et al. Contact lens-induced limbal stem cell deficiency. Ocular Surface. 2016;14(4):419-34. 9. Liesegang TJ. Physiologic changes of the cornea with contact lens wear. CLAO J. 2002;28:12-27. 10. Hickson S, Papas E. Prevalence of idiopathic corneal anomalies in a non-contact lens wearing population. Optom Vis Sci. 1997;74:293-7. 11. Stapleton F, Keay L, Jalbert I, Cole N. The epidemiology of contact lens related infiltrates. Optom Vis Sci. 2007;84:257-72. 12. Morgan PB, Efron N, Brennan NA, et al. Risk factors for the development of corneal infiltrative events associate with contact lens wear. Invest Ophthalmol Vis Sci. 2005;46:3136-43. 13. Szczotka-Flynn L, Ying J, Raghupathy S, et al. Corneal inflammatory events with daily silicone hydrogel lens wear. Optom Vis Sci. 2014;91:3-12. 14. Morgan PB, Efron N, Maldonado-Codina C, Efron S. Adverse events and discontinuations with rigid and soft hyper Dk contact lenses used for continuous wear. Optom and Vis Sci. 2005;82:528-35. 15. Lin MC, Yeh TN, Graham AD, et al. Ocular surface health during 30-day continuous wear: Rigid gas-permeable versus silicone hydrogel hyper-02 transmitted contact lenses. Invest Ophthalmol Vis Sci. 2011;52:3530-8. 16. Weissman BA. Corneal oxygen: 2015. CL Spectrum. 2015;30:25-29, 55. 17. Fleiszig SM. The pathogenesis of contact lens-related keratitis. Optom Vis Sci. 2006;83:E866-73. 18. Szczotka-Flynn L, Diaz M. Risk of corneal inflammatory events with silicone hydrogel and low Dk hydrogel extended contact lens wear: A meta-analysis. Optom Vis Sci. 2007;84(4):247-56 19. Stapleton F, Keay L, Edwards K, et al. The incidence of contact lens-related microbial keratitis in Australia. Ophthalmology. 2008;115:1655-62. 20. Liesegang TJ. Contact lens-related microbial keratitis: Part I: Epidemiology. Cornea. 1997;16:125-31. 21. Polse KA.Tear flow under hydrogel contact lenses. Invest Ophthalmol Vis Sci. 1979;18:409-13. 22. Ladage PM, Yamamoto K, Ren DH, et al. Effects of rigid and soft contact lens daily wear on corneal epithelium, tear lactate dehydrogenase, and bacterial binding to exfoliated cells. Ophthalmology. 2001;108:1279-88. 23. Zimmerman AB, Richdale K, Mitchell GL, et al. Water exposure is a common risk behavior among soft and gas-permeable contact lens wearers. Cornea. 2017;36:995-1001. 24. Cope JR, Collier SA, Schein OD, et al. Acanthamoeba keratitis among rigid gas permeable contact lens wearers, United States, 2005-2011. Ophthalmology. 2016;123(7):1435-41. 25. Seal DV, Bennett ES, McFadyen AK, et al. Differential adherence of Acanthamoeba to contact lenses: Effects of material characteristics. Optom Vis Sci. 1995;72(1):23-8. |


