The How and Why of Contact Lens Deposits
Optometrists need a comprehensive understanding of this complication to help patients avoid it.
By Heidi Wagner, OD, MPH
 |
Release Date: May 15, 2020
Expiration Date: May 15, 2023
Estimated time to complete activity: 2 hours
Jointly provided by Postgraduate Institute for Medicine (PIM) and Review Education Group.
Educational Objectives: After completing this activity, the participant should be better able to:
- Discuss the underlying mechanisms of contact lens deposits.
- Identify contact lens deposits in their patients.
- Recommend changes to reduce deposits in their contact lens wearers.
- Factor in lens material choices to improve comfort and vision.
- Describe how lens care options and surface treatments impact deposition.
Target Audience: This activity is intended for optometrists engaged in the care of patients with contact lens deposits.
Accreditation Statement: In support of improving patient care, this activity has been planned and implemented by the Postgraduate Institute for Medicine and Review Education Group. Postgraduate Institute for Medicine is jointly accredited by the Accreditation Council for Continuing Medical Education, the Accreditation Council for Pharmacy Education, and the American Nurses Credentialing Center, to provide continuing education for the healthcare team. Postgraduate Institute for Medicine is accredited by COPE to provide continuing education to optometrists.
Faculty/Editorial Board: Heidi Wagner, OD, MPH, Ohio State University
Credit Statement: This course is COPE approved for 2 hours of CE credit. Course ID is 67923-CL. Check with your local state licensing board to see if this counts toward your CE requirement for relicensure.
Disclosure Statements:
Dr. Wagner has contracted research with Alcon and has received honoraria from Wink Productions.
Managers and Editorial Staff: The PIM planners and managers have nothing to disclose. The Review Education Group planners, managers and editorial staff have nothing to disclose.
In this era of disposability, many eye care providers are less concerned about contact lens deposits. In 2019, daily disposable soft contact lenses (SCLs) accounted for 35% of international lens prescribing and 44% of lenses prescribed in the United States.1 As the market share of conventional and planned replacement SCLs shrinks, lens deposits may be less prevalent and less severe; still, lens deposition remains a factor, especially with the expanded use of specialty contact lenses.
Specialty SCLs, gas permeable (GPs) lenses and hybrids play an important role in the United States market of 45 million contact lens wearers.2 Specialty SCLs and hybrids are typically replaced far less frequently than daily disposables—often at quarterly or six-month intervals. In contrast to SCLs, GPs are often replaced “reactively” (i.e., when the patient requires a change in lens power or experiences reduced comfort, degraded vision or lens loss or damage) rather than on a planned schedule.
Contact lens deposits significantly impact the patient’s lens wearing experience and ocular health. Lens spoilage can potentially reduce lens surface wettability and adversely impact patient comfort, wearing time and quality of vision. Further, lens deposits can result in contact lens-related ocular pathology, including papillary conjunctivitis, punctate keratitis, corneal inflammatory events and even microbial keratitis.3,4 This article reviews how to identify various types of lens deposits, describes the impact of lens material choices on comfort and vision and delineates how lens care options and surface treatments impact deposition.
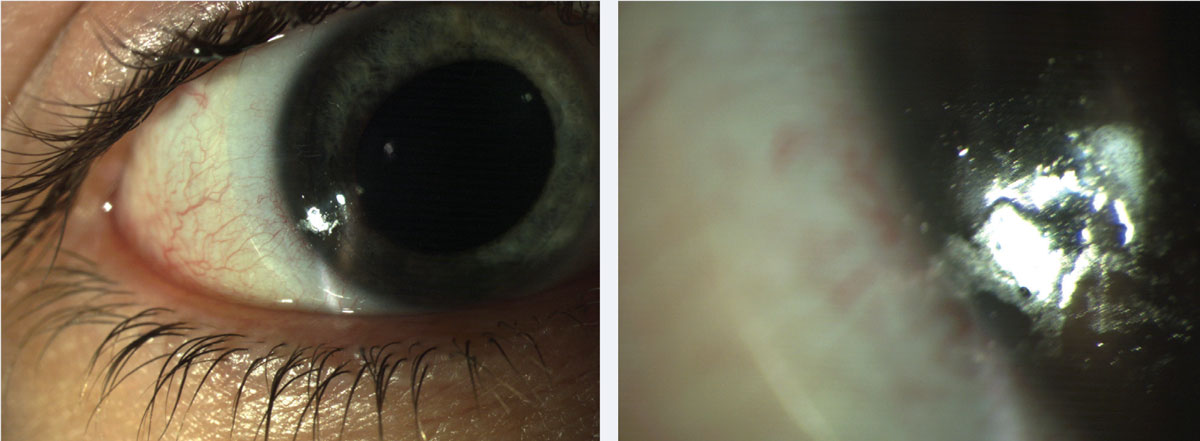 |
| Fig. 1. Patients can present with both protein and lipid deposits. These common lens deposits are shown here at (left) low and (right) high magnification. Click image to enlarge. |
Underlying Mechanisms
A general understanding of the underlying mechanisms of lens deposits and an awareness of strategies to reduce them remain integral to contemporary contact lens practice. Lens depositing is influenced by many factors, including patient compliance, individual tear chemistry and environment. Individual tear chemistry is evidenced by lipid composition, protein profile, mucin and electrolyte analysis—characteristics that manifest in the wearer response.5 Understanding these interactions can help the eye care provider optimize lens performance and minimize adverse events.
Identifying Deposits
Lens deposition begins within minutes of wear.6 While surface deposits may be minimized by increasing the frequency of lens replacement, variation exists among individual patients with regards to tear chemistry and compliance with the lens care regimen.7 Practitioners must be vigilant in identifying lens deposits with all types of lens materials and replacement regimens.
Contact lens deposits are best identified through observation of the lens on the eye with biomicroscopy under varying illumination and magnification. A lens loupe is a practical alternative, particularly if the lens is damaged and could potentially harm the eye.
Lens deposits can be distinguished by color, structure and location. Identification of the predominant deposit can guide the practitioner in management decisions. There are a number of common types of deposits practitioners should be aware of.
Proteins and lipids. These are long-recognized lens deposits in contact lens practice.8,9 Protein deposits occur as lysozyme binds to the lens surface and undergoes structural changes that impair its function. These changes, termed protein denaturation, are influenced by numerous factors such as the lens substrate, pH and temperature.6 Protein deposits are characterized by an opaque film on the lens that becomes more obvious over time. In contrast, lipid deposition is characterized by a shiny, lubricious appearance. Both are present in SCLs and GPs. Protein and lipid deposits can be observed in combination in an individual patient (Figure 1).
Lens calculi. Sometimes referred to as “jelly bumps,” these deposits are distinct, localized elevations on the anterior surface of the SCL. Lens calculi are composed of calcium, lipid and mucoprotein inherent in the tear film (Figure 2).10 Their formation is attributed to depletion of the aqueous tear layer that results in a hydrophobic zone that, in turn, promotes deposition.11 If significant in number and size, they can degrade comfort and vision.
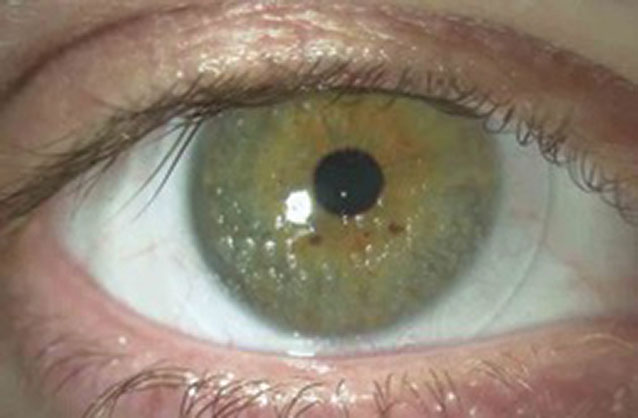 |
| Fig. 2. This patient presented with distinct, localized elevations on the anterior surface of the lens—lens calculi. Click image to enlarge. |
While frequently observed in the era of conventional SCL lens wear, they are relatively uncommon in lenses that are replaced monthly or more frequently. Thus, if practitioners observe lens calculi in patients wearing lenses with shorter replacement cycles, wearers may be “stretching” their replacement cycles. As the deposit is embedded within the matrix of the lens, replacement is necessary.
This type of deposit is more commonly observed in high-water, ionic (group IV), hydrogel lens materials.11 The practitioner can address this problem by refitting the patient into a different lens material, though simply reinforcing the lens replacement schedule or refitting into daily disposable contact lenses may address the problem.
Fungal deposits. Given the predilection of fungus for lens materials of a higher water content, this type of deposit, which is often characterized by a filamentary appearance, is more commonly observed in soft or hybrid lenses.12
Fungal deposits may be associated with poor disinfection regimens. This can occur when patients use saline instead of a multipurpose disinfection solution or when part-time wearers or multiple pair (e.g., colored lenses) wearers store lenses in solution for extended periods of time. Additionally, patients who disinfect lenses with hydrogen peroxide systems may be unaware that the neutralized disinfection solution is saline and that the solution must be replaced every seven days if the lenses are not worn.13 Therefore, it is important to prescribe a lens care system that is appropriate for the patient’s wearing schedule and ensure that the patient understands how to use it.
Iron deposits. Characteristically round and brown-to-orange in color, such deposition may be a consequence of incorporating tap water into the lens care regimen, despite published evidence of the association of Acanthamoeba with water exposure.14 In a survey of more than 1,000 SCL wearers, 31% reported rinsing their SCLs with tap water on at least one occasion, and 10% reported always or fairly often rinsing their lenses with tap water.15 Of the wearers who reported rinsing their lenses with tap water, 41% reported also storing their lenses in tap water.15
Upon identifying iron deposits, eye care providers should emphasize that no amount of water exposure is acceptable. This message may be reinforced by promotional materials, such as the “no water” stickers distributed by the Cornea, Contact Lenses and Refractive Technologies Diplomate Section of the American Academy of Optometry (Figure 3).
 |
| Fig. 3. These “no water” stickers, distributed by the American Academy of Optometry, can help reinforce to patients that no amount of water exposure is acceptable. Click image to enlarge. |
Mucin balls. These deposits are round, semitransparent spheres ranging in size between 40µm and 120µm. While mucin balls have been observed in a variety of lens materials, they are more frequently associated with silicone hydrogels (SiHy). Research suggests that their formation is based on a mechanical interaction between the cornea and high modulus SiHy materials.16
Mucin balls do not appear to impact vison or comfort and, therefore, can easily be differentiated from other types of lens deposits. They are more likely observed in first-generation SiHy products that are characterized by “stiffer” (high modulus) lens materials.
Environmental debris. Make-up, such as mascara and eyeliner, is a common source of lens deposits. While eye make-up may be easily identified by color and texture, identifying the source of other contaminants degrading the lens surface may prove to be more elusive.17
Lotions transferred from fingertips and aerosol hairspray can also bind to the lens. These types of deposits can be eliminated by proper hand washing before lens handling and applying make-up after lens insertion (Figure 4).
Other potential sources of environmental lens deposits include organic debris such as leaf litter and inorganic contaminants such as a metallic foreign body. If you suspect a metallic foreign object, always perform a more extensive eye examination, given the possibility of an intraocular foreign body.
Lipid, protein and exogenous contaminants are likely to deposit on both GP and SCL lens materials. Unique to GP lenses, however, is poor wetting exhibited in newly dispensed lenses. This is somewhat less common as water-soluble products currently used in the manufacturing process have largely eliminated the oily residue (i.e., “pitch”) that was previously part of the manufacturing process.
This problem can generally be solved by plasma cleaning or soaking lenses in an appropriate conditioning solution prior to dispensing. Lens cleaners can also be used with appropriate materials, as discussed below.
On occasion, topical and systemic medications have been associated with lens discoloration in SCLs. For example, rifampin, a drug used to treat tuberculosis, can cause an orange discoloration of contact lenses.18 A similar phenomenon has been reported with sulfasalazine, which is used to manage inflammatory bowel disease.19 Lens discoloration, ranging from pink to brown, has also been observed with some topical medications, such as the epinephrine ophthalmic drops used in the past to treat glaucoma.
While these conditions are not observed in every day clinical practice, the practitioner should be aware of the potential of oral and topical medications to influence the tear ocular environment.20
Lysozyme deposits. Notably, lysozyme deposition may provide beneficial effects during contact lens wear, as lysozyme exhibits antibacterial and anti-inflammatory properties.6 Research also shows that lactoferrin in the tears has the potential to work in concert with lysozyme to inhibit gram-positive and gram-negative bacteria.6 However, further study is needed to better understand these interactions.
Lens materials influence the deposition of tear-derived products that, in turn, influences lens comfort.21 Some investigators have also challenged the belief that lens deposition negatively impacts comfort, noting that lysozyme has, on occasion, been associated with increased comfort in HEMA-based lens materials. This was attributed to the fact that lysozyme retains a higher degree of activity when deposited on traditional hydrogel lens materials compared with silicone hydrogels.22 They propose the development of lens materials that can selectively bind “good” deposits and inhibit “bad” deposits.23
Lens Material
The FDA classifies hydrogel contact lenses as ionic (groups III and IV) and nonionic (groups I and II). Groups II and IV exhibit a higher water content (≥50% water) than groups I and III. SiHy, in general, are characterized by lower water content but higher oxygen permeability.6
The rate of protein deposition is significantly related to the lens material. Polymethyl methacrylate (PMMA) and SiHy lens materials deposit less lysozyme than hydrogels, and lysozyme is particularly prevalent in high-water, ionic lens (group IV) materials.24 The external environment and lens handling further expose the lenses to contaminants.
SCLs provide an ideal medium to attract lens deposits, given the hydrophilic surface. Hydrogel lenses contain methacrylic acid to increase water content and oxygen permeability.25 Consequently, HEMA-based lens materials exhibit a predisposition toward protein deposition, as the negatively charged methacrylic acid binds to positively charged proteins, including lysozyme.26 Thus, refitting patients wearing SCLs from high-water, ionic lens (group IV) materials to low-water, non-ionic lens (group I) materials may reduce protein deposits. SiHy lenses, while highly oxygen permeable, are potentially hydrophobic in nature. They may exhibit reduced wettability and a greater tendency towards lipid deposition compared with their HEMA-based counterparts.27
Rigid lens materials exhibit a parallel story. All but obsolete, PMMA contact lenses were deposit resistant but impervious to oxygen. GP lenses are permeable to oxygen in varying degrees based on the polymer components. Silicone was added to the lens material to create silicone acrylate (SA) lens materials. This resulted in an increased oxygen permeability but more protein deposition.
Fluorine was then added to maintain oxygen permeability and improve wettability of the current generation of fluorosilicone acrylate (FSA) lens materials. Earlier generation SA lenses tended to deposit proteins while newer FSA lenses tend to deposit lipids.28
Given these various material characteristics, clinicians should customize the lens material to the individual patient. For example, a hyper-Dk lens material may be desirable for overnight wear in orthokeratology while a moderate-Dk lens material may be ideal for a patient who tends to deposit lipids.
In addition, the provider can further tailor the lens care regimen to the needs of the lens wearer. For example, a heavy lipid depositor who also requires a high-Dk lens material could benefit from a more rigorous lens care system as described below.
| Other Considerations Further consideration for giant papillary conjunctivitis (GPC) is warranted, given its association with lens deposits. GPC is most commonly associated with SCLs but can be associated with GPs, as well as sutures following surgery.3 While the condition was initially described as a “reaction” to soft contact lenses, the term was later redefined by researchers who postulated that the syndrome was immunologic in origin with deposits on the lenses serving as an antigen (type IV immune response).35 A mechanical component has also been suggested, although there is no consensus on this issue.36 GPC is characterized by papillae on the tarsal conjunctiva. In mild cases, patients may have symptoms of lens awareness. In severe cases of GPC, patients may experience excessive lens movement, substantial lens depositing and lens intolerance. Contact lens-induced GPC can be managed by increasing lens replacement frequency, decreasing lens wearing time or changing lens materials. Concurrent pharmacological management, such as mast-cell inhibitors/antihistamine combination drugs and topical steroids, can be added. |
Lens Wear and Care
Proper contact lens wear and care practices are essential for all contact lens modalities, and they should be tailored to the particular lens modality and patient. In a recent survey administered by the Centers for Disease Control and Prevention, six of seven contact lens wearers acknowledged at least one behavior that places them at risk for a contact lens-related adverse event.2 Eye care providers play an important role in educating all contact lens wearers at the initial fitting as well as reinforcing best practices at every follow-up visit.
Clinicians should provide specific guidance based on the unique needs of the patient, including the lens material, replacement schedule, contact lens care, tear chemistry and history of compliance.
SCLs. Appropriate lens care goes a long way in maintaining a clean lens surface. Chemical disinfection systems (commonly designated as multipurpose solution [MPS]) combine cleaning, rinsing and disinfection. While MPS is integral to lens care, it is useful to remember that its success is based on its ability to deliver key components of the lens care regimen: cleaning, rinsing, disinfecting and storage.
Cleaning removes loosely adhered deposits, as does lubrication. Rinsing removes the debris and avoids the introduction of additional external contaminants. Proper disinfection and storage limits microbial intrusion. It is important that patients remember that all lens care components—including the lens case, when applicable—are part of the lens care system.
In studies where the FDA required the manufacturers to inoculate the lenses with one million organisms to study the efficacy of a lens care system, the inclusion of a cleaning step removed one log unit of microorganisms from the lens. If the cleaner was rinsed from the lens, two additional log units of microorganisms were further eliminated.17
This work reinforces the need for digital cleaning, even with MPS. The FDA further discouraged the promotion of “no rub” lens care systems after the voluntary removal of two lens care products from the marketplace following their association with Fusarium and Acanthamoeba.29-31 Further studies have supported digital rubbing and rinsing to minimize deposits and limit bacterial contamination in reusable soft and GP lenses.32,33
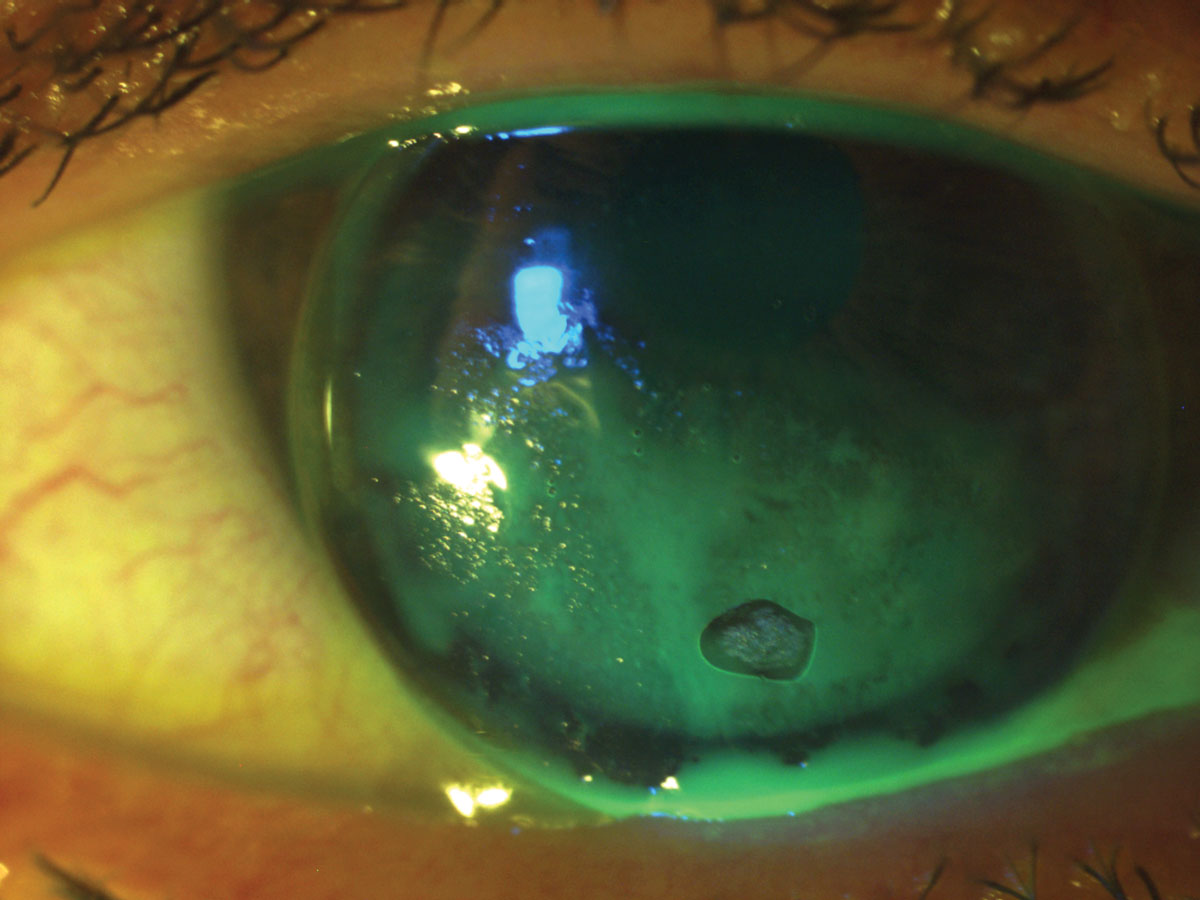 |
| Fig. 4. In this case, lip balm was inadvertently transferred to the lens, resulting in poor GP lens wetting. Click image to enlarge. |
A separate surfactant or enzymatic cleaner is rarely indicated for two-week or monthly replacement SCLs, although these products may be added to the care regimen for “heavy depositors.” Surfactant cleaners remove loosely adhered lens debris, unbound protein and microbial contamination. As these cleaners are less accessible than in the past, patients may require additional direction regarding where to purchase them.
Hydrogen peroxide systems are a particularly effective preservative-free disinfection option. Contemporary systems contain a surfactant and, in one system, a wetting agent. However, anecdotal reports suggest lens residue may be associated with solutions that contain a proprietary wetting agent. This can be resolved by switching to another hydrogen peroxide product without the wetting agent.
Practitioners should be cognizant of current MPS systems, make an initial prescribing decision and modify as needed. They should also be alerted to potential patient pitfalls, such as “topping off” (which can reduce disinfection efficacy), purchasing alternate products and not completing the cleaning regimen as directed. It should also be noted that SCL wearers who have an ample supply of lenses are more likely to replace their lenses at recommended intervals.34
GPs. One-bottle care systems for cleaning, rinsing, disinfection and storage are also available for GP lenses. As with MPS SCL solutions, digital cleaning can enhance the efficacy of the process. For example, Unique pH (Menicon) and Boston Simplus (Bausch + Lomb) provide one-bottle convenience. Two-bottle systems, such as Boston Original and Advance (Bausch + Lomb), incorporate a separate abrasive cleaner that enhances the cleaning regimen. Boston Original was designed for lower-Dk SA lenses, which tend to deposit proteins, while Boston Advance was developed for higher-Dk FSA lens materials that deposit lipids.
Clinicians can also manage lens deposits by being judicious in the addition of Hydra-PEG (Tangible Science) and surface treatments. Hydra-PEG is a biocompatible polymer that may be applied to GP or hybrid lenses as part of the manufacturing process. As described by the manufacturer, the coating promotes a lubricious lens surface that is designed to inhibit lens deposits and fogging. Tangible Clean (Tangible Science) is an MPS solution designed for Hydra-PEG coated lenses. It can also be used for uncoated lenses.
Abrasive cleaners are contraindicated in plasma-treated lenses, hyper-Dk lens materials and with Hydra-PEG. Non-abrasive cleaners that contain alcohol are particularly effective with lipid removal and are compatible with hyper-Dk lens materials; however, no consensus exists regarding their use with plasma-treated lenses, and they are contraindicated with Hydra-PEG.
Given that tap water is contraindicated with all contact lenses, low viscosity solutions such as saline or MPS should be employed to rinse the cleaner from the lens. As this inadvertently introduces a third step, one-step hydrogen peroxide systems provide a practical alternative whereby the disinfection solution also contains a surfactant cleaner and the solution neutralizes to saline.
Anecdotally, patients who successively use MPS systems with corneal GPs may require a more rigorous system with scleral lenses, presumably because corneal lenses exhibit more tear exchange. Heavy depositors may also benefit from periodic cleaners such as enzymatic cleaners that remove protein or Progent (Menicon), which exhibits both cleaning and disinfection properties. Progent may be used as frequently as every two weeks for heavy depositors and can be used in office. Patients who experience difficulty digitally cleaning their lenses because of the lens geometry (e.g., lenses for keratoconus with steep base curves) may also benefit from incorporating periodic cleaners.
Contact lens deposits are a well-known clinical challenge. This challenge can lead to reduced comfort and vision and negatively impact ocular health. Often, changing the contact lens or care regimen is not enough to ward off deposits. Many factors impact a patient’s chances of experiencing this complication, including the lens material, surface treatments, wear schedules, care regimens and the patient’s individual tear fluid composition.
1. Morgan PB. International contact lens prescribing in 2019. Contact Lens Spectrum. 2020;35(1):26-32. 2. Cope JR, Collier SA, Nethercut H, et al. Risk behaviors for contact lens–related eye infections among adults and adolescents—United States, 2016. MMWR Morb Mortal Wkly Rep. 2017;66:841-5. 3. Allansmith MR, Korb DR, Greiner JV, et al. Giant papillary conjunctivitis in contact lens wearers. Am J Ophthalmol. 1977;83(5):697-708. 4. Aswad MI, John T, Barza M, et al. Bacterial adherence to extended wear soft contact lenses. Ophthalmology. 1990;97(3):296-302. 5. Mann A, Tighe B. Contact lens interactions with the tear film. Exp Eye Res. 2013;117(12):88-98. 6. Omali NB, Subbaraman LN, Coles-Brennan C, et al. Biological and clinical implications of lysozyme deposition on soft contact lenses. Optom Vis Sci. 2015;92(7):750-7. 7. Ilhan B, Irkec M, Orhan M, et al. Surface deposits on frequent replacement and conventional daily wear soft contact lenses: a scanning electron microscopic study. Clao J. 1998;24(4):232-5. 8. Lin ST, Mandell RB, Leahy CD, et al. Protein accumulation on disposable extended wear lenses. CLAO J. 1991;17(1):44-50. 9. Jones L, Evans K, Sariri R, et al. Lipid and protein deposition of N-vinyl pyrrolidone-contacting group II and group IV frequent replacement contact lenses. CLAO J. 1997;23(2):122-6. 10. Begley CG, Waggoner PJ. An analysis of nodular deposits on soft contact lenses. J Am Optom Assoc. 1991;62(3):208-14. 11. Hart DE, Tidsale RR, Sack RA. Origin and composition of lipid deposits on soft contact lenses. Ophthalmology. 1986;93(4):495-503. 12. Chen J, Fraser T, Fisher D, et al. Characteristics of fungal growth in soft contact lenses. Int Contact Lens Clin. 2000;26(4):84-91. 13. Kilvington S, Lam A, Nikolic M, et al. Resistance and growth of Fusarium species in contact lens disinfectant solutions. Optom Vis Sci. 2013;90(5):430-8. 14. Radford CF, Minassian DC, Dart JK. Acanthamoeba keratitis in England and Wales: incidence, outcome, and risk factors. Br J Ophthalmol. 2002;86(5):536-42. 15. Zimmerman AB, Richdale K, Mitchell GL, et al. Water exposure is a common risk behavior among soft and gas-permeable contact lens wearers. Cornea. 2017;36(8):995-1001. 16. Tan J, Keay L, Jalbert I, et al. Mucin balls with wear of conventional and silicone hydrogel contact lenses. Optom Vis Sci. 2003;80(4):291-7. 17. Weisbarth R, Henderson B. Hydrogel Lens Care Regimens and Patient Education. In: Bennett ES, Weissman BA, eds. Clinical Contact Lens Practice. Philadelphia, PA: Lippincott Williams & Wilkens; 2005. 18. Lyons RW. Orange contact lenses from rifampin. N Engl J Med. 1979;300(7):372-3. 19. Krezanoski JZ. Topical medications. Int Ophthalmol Clin. 1981;21(2):173-6. 20. Miller D, Brooks SM, Mobilia E. Adrenochrome staining of soft contact lenses. Ann Ophthalmol. 1976;8(1):65-67. 21. Jones L, Brennan NA, Gonzalez-Meijome J, et al. The TFOS international workshop on contact lens discomfort: report of the contact lens materials, design, and care subcommittee. Invest Ophthalmol Vis Sci. 2013;54(11):TFOS37-70. 22. Subbaraman LN, Glasier MA, Varikooty J, et al. Protein deposition and clinical symptoms in daily wear of etafilcon lenses. Optom Vis Sci. 2012;89:1450-9. 23. Subbaraman LN. Is contact lens deposition good or bad? Contact Lens Update. 2016. https://contactlensupdate.com/2016/06/25/is-contact-lens-deposition-good-or-bad. 24. Subbaraman LN, Glasier MA, Senchyna M, et al. Kinetics of in vitro lysozyme deposition on silicone hydrogel, PMMA, and FDA groups I, II, and IV contact lens materials. Curr Eye Res. 2006;31(10):787-96. 25. Tighe B. Rigid lens materials. In: Efron N, ed. Contact Lens Practice. Oxford, United Kingdom: Butterworth-Heinemann; 2010. 26. Jones L, Mann A, Evans K, et al. An in vivo comparison of the kinetics of protein and lipid deposition on group II and group IV frequent-replacement contact lenses. Optom Vis Sci. 2000;77(10):503-10. 27. Nicolson PC, Vogt J. Soft contact lens polymers: an evolution. Biomaterials. 2001;22(24):3273-83. 28. Bennett ES, Henry VA, eds. Clinical Manual of Contact Lenses. 4th ed. Philadelphia: Lippincott Williams & Wilkins; 2013. 29. Centers for Disease Control and Prevention. Update: Fusarium keratitis--United States, 2005-2006. MMWR Morb Mortal Wkly Rep. 2006;55(20):563-4. 30. Joslin CE, Tu EY, Shoff ME, et al. The association of contact lens solution use and Acanthamoeba keratitis. Am J Ophthalmol. 2007;144(2):169-80. 31. Food and Drug Administration. FDA Letter to Firms with Marketing Clearance for No-rub Multipurpose Contact Lens Solutions. 2019. www.fda.gov/medical-devices/contact-lenses/fda-letter-firms-marketing-clearance-no-rub-multipurpose-contact-lens-solutions. 32. Cho P, Poon HY, Chen CC, et al. To rub or not to rub? - effective rigid contact lens cleaning. Ophthalmic Physiol Opt. 2020;40(1):17-23. 33. Cho P, Cheng SY, Chan WY, et al. Soft contact lens cleaning: rub or no-rub? Ophthalmic Physiol Opt. 2009;29(1):49-57. 34. Schnider C. The ‘Pantry Load’ Effect – Can it help drive more compliant contact lens replacement? Optom Vis Sci. 2012;89:E-abstract 120652. 35. Spring TF. Reaction to hydrophilic lenses. Med J Aust. 1974;1(12):449-50. 36. Donshik PC, Ehlers WH, Ballow M. Giant papillary conjunctivitis. Immunol Allergy Clin North Am. 2008;28(1):83-103, vi. |


