Neurotrophic keratitis (NK) is a potentially sight-threatening condition marked by decreased or absent corneal sensation. The condition is caused by damage both to the trigeminal nerve along its corneal distribution and at any point in the pathway from the ganglion to the basal plexus. This damage compromises corneal integrity by altering the metabolism and mitosis processes of the corneal epithelium, which results in reduced innate immunity of the ocular surface and delayed healing.1,2 If allowed to progress, damage could advance to the point of ulceration and, in severe cases, corneal perforation.1-3
NK is easy to manage early on in the disease process but remains one of the most challenging corneal conditions to treat in its severe stages. This article will review the causes, diagnostic findings and treatments available for NK throughout all stages of disease.
Trace it to the Source
The most common etiologies of NK are herpes simplex virus (HSV) and varicella zoster virus (VZV).1 Among herpes viruses, zoster causes more severe neurotrophy, as it may inflict damage both centrally at the ganglion and peripherally at the basal plexus. Alternatively, HSV only causes peripheral nerve damage. Patients suffering from herpes zoster ophthalmicus experience shorter corneal nerve length and lower corneal nerve count.4 The zoster virus lies dormant in the trigeminal ganglion, and when it reactivates along the nasociliary distribution, it results in corneal compromise.4 In both HSV and VZV, these damaging effects occur quickly and have long-lasting impacts on the integrity of the corneal epithelium.
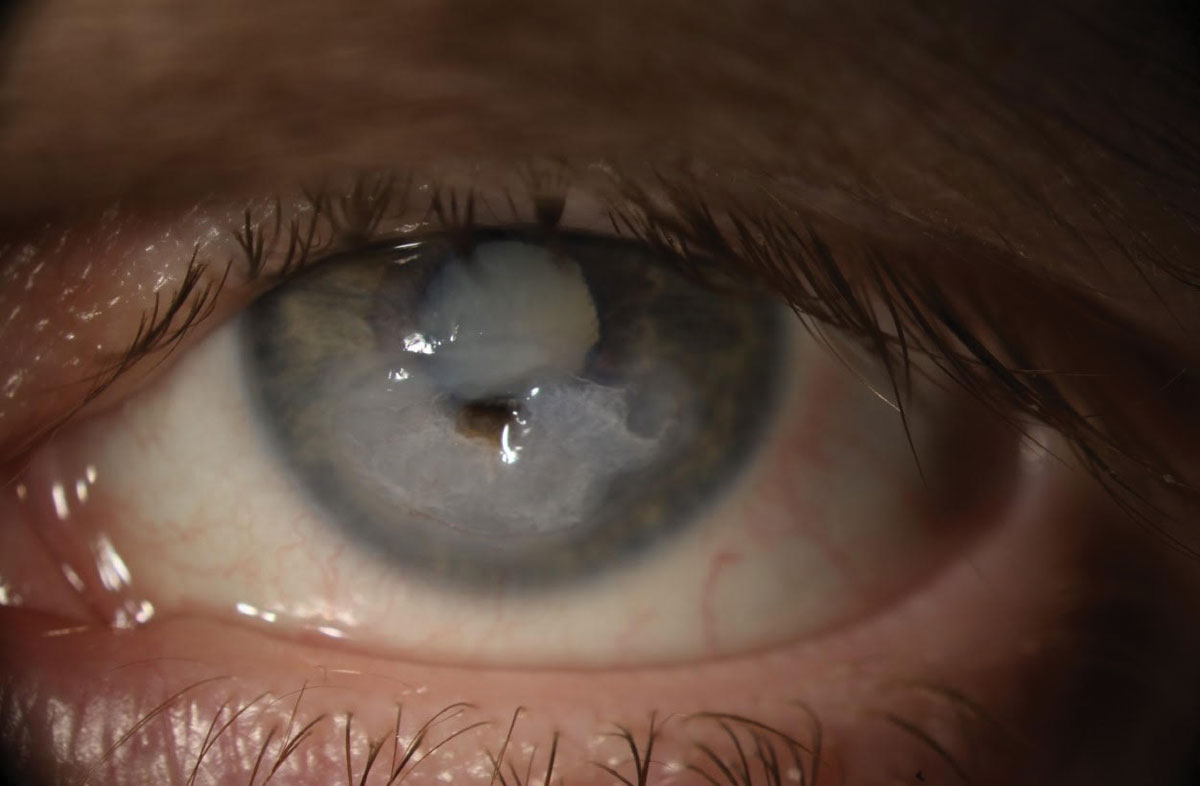 |
| Fig. 1. A 51-year-old Caucasian female presented with progressively worsening vision in the left eye over the last two months but no more discomfort. She reported using bacitracin-Polymyxin B- neomycin-hydrocortisone ophthalmic solution TID and diclofenac BID for approximately two months while in the hospital. Click image to enlarge. |
Beyond viral eye disease, there are a variety of other causes of NK, including repeated corneal surgeries, chemical burns, contact lens abuse, medications, tumors and surgical complications resulting in trigeminal nerve palsies.1,5
Systemic disorders such as diabetes also contribute to NK. The longer a patient has diabetes, the more severe their corneal nerve damage might be. This manifests in diminished corneal tissue sensation similar to that occurring with peripheral neuropathy. Additionally, peripheral treatment with panretinal photocoagulation for proliferative diabetic retinopathy worsens the degree of neurotrophy.5 In fact, corneal confocal microscopy has been shown to be useful in the early diagnosis of peripheral neuropathy due to its ability to detect subtle reductions in corneal nerve density and length.6 This form of microscopy can assess small nerve fibers, which neuropathy affects first.6 Because of the link between diabetic neuropathy and corneal integrity, consider the role of underlying neurotrophy in patients with diabetes and other ocular surface disease.
Regardless of its specific source, corneal neurotrophy is typically not associated with pain or discomfort due to the lack of sensation it is accompanied by. These patients will, however, likely report reduced vision, especially as the condition becomes more severe. Medicamentosa is a potential cause of corneal neurotrophy that should always be considered as a differential and managed appropriately. Topical medications most likely to have noxious ocular surface effects include anesthetics, beta-blockers and non-steroidal anti-inflammatories (NSAIDs). Most of these medications use benzalkonium chloride as the preservative.7 Monitor the ocular surface closely in glaucoma patients using beta-blockers and in post-surgical patients using NSAIDs, as these are the medications most commonly prescribed to these patients.
Using a greater number of medications further increases the risk of toxicity.7 If topical toxicity is suspected, discontinue medications and monitor the cornea for improvement. Systemic medications have also been shown to perpetuate corneal neurotrophy. These include antihistamines, neuroleptics and antipsychotics.2 Consult primary care providers and consider drug cessation if these mediations are contributing to corneal compromise.
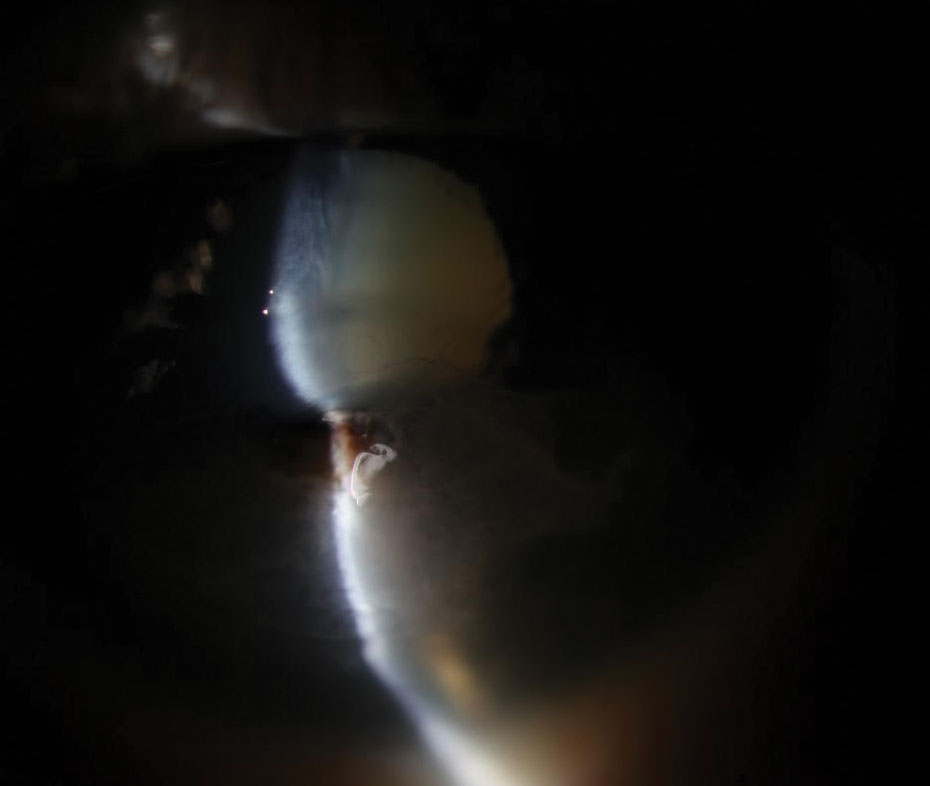 |
| Fig. 2. Slit lamp exam revealed a perforated corneal ulcer involving the inferior visual axis with a collapsed anterior chamber and iris prolapse in the left eye. Click image to enlarge. |
The Three Stages of NK
There are three traditional clinical stages of NK. The first involves tear dynamic alteration, punctate epithelial erosion, superficial neovascularization and stromal scarring. These findings are common and can be mistaken for other ocular surface diseases, but decreased corneal sensation, lack of symptoms and patient history will help with the diagnosis.1,2,8
The second stage of NK is marked by a persistent epithelial defect. The defect may be surrounded by a loose, edematous or boggy epithelium. Stromal edema and folds in Descemet’s membrane may also accompany these clinical signs. Cells and flare, which may occasionally worsen into a sterile hypopyon, are often present in this stage and can make the differentiation between microbial keratitis and neurotrophic disease more challenging.1,2,5,8
The third and final stage of neurotrophy involves corneal stroma ulceration. This is defined as an epithelial break with underlying stromal inflammation. The stroma is made up of organized collagen fibers called lamellae. Once the stroma is penetrated, the collagen fibrils start to degrade. This damage attracts white blood cells, which infiltrate the damaged tissue. These immune cells proceed to release matrix metalloproteinases (MMPs) and oxygen radicals, which are pro-inflammatory and cause the stroma to progressively degrade.9 This may lead to corneal melt and perforation—an ocular emergency that requires immediate attention and treatment to best preserve vision and globe integrity.1,2,8
Clinical Diagnosis
Diagnosing NK involves evaluating corneal sensation. Corneal aesthesia can be tested using a few different methods. Cotton wisps and (non-flavored) dental floss are used most frequently, as they are inexpensive and readily available.
Approach the unaffected eye first and test it in quadrants and centrally. Then, proceed to the other eye and test the same zones, allowing the patient to grade the sensitivity in comparison with its counterpart.
Relative neurotrophy is gradable using both subjective responses and observable differences in patient blink force and recoil. For consistency, use a near uniform length of wisp or floss each time you test (~3cm is standard) to ensure equal force is applied with each touch. Alternatively, the Cochet-Bonnet esthesiometer is a more repeatable and quantifiable test of aesthesia. It records a patient’s response to contact with a nylon line on a scale between 0cm and 6cm.10 A lower reading indicates more reduced corneal sensitivity.11
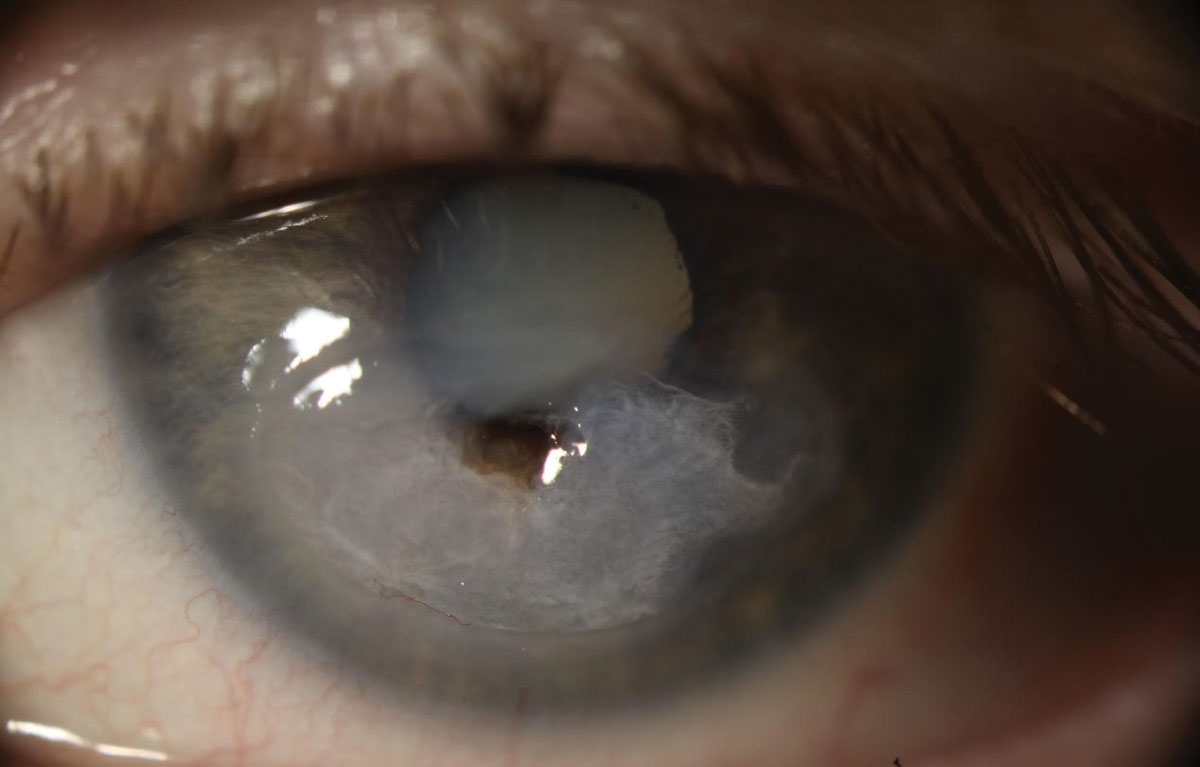 |
| Fig. 3. The patient underwent emergency penetrating keratoplasty (PK) followed by a repeat PK with cataract extraction. The culture performed prior to the first corneal transplant was negative for microbial growth. Due to a persistent epithelial defect following PK, she needed multiple Prokera (Bio-Tissue) membranes, tarsorrhaphy and an amniotic membrane transplant. Post-op visual acuity was 20/400 with pinhole acuity ranging from 20/80 to 20/200. Click image to enlarge. |
Treatment Strategy
Treatment of NK is based on disease etiology and severity. In stage one, optimizing the ocular surface is the primary goal. Preservative-free artificial tears, punctal occlusion and Restasis (Allergan) or Xiidra (Novartis) are frequently prescribed to improve tear volume. Comorbidities, such as meibomian gland dysfunction, exposure keratitis and limbal deficiency, should also be addressed to prevent further corneal damage.
When NK progresses to stage two, closure of the defect and prevention of progressive stromal thinning are the primary objectives. Use scleral lenses, bandage soft contact lenses and amniotic membranes in attempts to close the epithelial defect. Scleral lenses can act as a moisture chamber to offer a fluid reservoir and buffer against the mechanical force of the blink. Bandage contact lenses provide a short-term barrier to lid friction. Amniotic membranes contain placental tissue and, thus, a variety of growth factors. They have been shown to increase corneal epithelialization, promote tissue repair and heal persistent epithelial defects.12 Additionally, they decrease MMPs, which normally reduce tissue growth and turnover and, thus, inflammation.13 Each of these therapies should be combined with antimicrobial prophylaxis to prevent bacterial super-infection.
Consider a temporary lateral tarsorrhaphy to heal persistent defects. A temporary tarsorrhaphy joins the eyelids together with three or four sutures. Wait until the wound resolves to release the tarsorrhaphy, as premature suture removal makes these epithelial defects more likely to recur.8,14 Two alternatives to a tarsorrhaphy are a Botox injection into the levator to create a temporary ptosis and palpebral spring use.8,15 These surgical interventions may be helpful in cases of pure exposure keratopathy as well.
Serum-based eye drops are becoming more widely used therapies for stages one and two of NK. These drops are created from blood samples, which are clotted, centrifuged and diluted with either sterile saline or a balanced salt solution.16 All dilution preparations have proved useful in resolving persistent epithelial defects.17 Additionally, umbilical cord serum, substance P with insulin-like growth factor 1 and nerve growth factor drops are being explored as future mainstream NK treatments due to their ability to repair nerve damage.2,8,11,18,19 In fact, Oxervate (Dompé)—a recombinant human nerve growth factor—was recently FDA-approved for the treatment of NK. Treatment includes six daily drops over an eight-week course and has shown extremely promising results in repairing persistent corneal defects.20 Nerve growth factor promotes epithelial healing and increases corneal sensitivity.21,22
Also gaining popularity is corneal neurotization, which restores corneal sensation by transferring healthy nerve tissue to the limbus of the affected cornea. It is most commonly performed on the distal ends of the supratrocheal and supraorbital nerves.23,24 The donor nerve is drawn together with the damaged nerve with the hope of regenerating corneal sensation, which could take up to six months following surgery.23 Although neurotization procedures are extensive, the least invasive techniques use cadaver nerve tissue or endoscopy.23 Neurotization is thought to lead to direct nerve sprouting and provide a potential cure to NK.23
Topical steroids may be helpful in managing neurotrophy caused by chemical burns; however, due to their immunosuppression and delayed healing effects, they are considered controversial in treating pure NK. Further, they have been found to increase the risk of corneal melting and perforation by up-regulating collagenases, which leads to enhanced stromal breakdown. In an effort to prevent this, avoid steroids if possible.1,2,25 If a steroid is necessary, pair it with an appropriate antibiotic to keep normal flora from infecting the NK.
Stage three of NK is characterized by a sterile ulceration or melting of the stroma with the potential to perforate. It is promoted by the prolonged presence of epithelial defects. Collagenase activity is up-regulated by the presence of these chronic epithelial defects. This leads to thinning of the stroma with subsequent scarring and warpage of the cornea if re-epithelialization occurs or, worse— in the absence of re-epithelialization—if progression to perforation takes place.5
While steroids are generally avoided due to their ability to potentiate collagenase activity, medications that down-regulate these enzymes can be helpful by decreasing potentiators of melt. These include N-acetylcysteine, oral tetracycline antibiotics and medroxyprogresterone.1,9,25
If perforation is impending or has already occurred, treatment takes on a new level of urgency. Treatments include cyanoacrylate glue or fibrin adhesive application and amniotic membrane transplantation as temporizing measures. Adhesive options are therapeutic via tectonic improvement and polymorphonuclear leukocyte reduction, which arrests stromal lysis via the inhibition of collagenases.15,25,26 If ulceration continues to worsen, follow this step with a more definitive conjunctival flap or keratoplasty.
Conjunctival flaps are a definitive treatment for neurotrophy but come at a significant burden to visual potential. They promote healing by providing fibrovascular tissue rich in growth factors. The primary goal is to preserve globe integrity. Conjunctival tissue is not susceptible to the same mechanisms that lead to neurotrophic disease and, thus, the potential for perforation is eliminated. Flaps are reserved for chronic ulcers with poor visual prognoses because although the conjunctival flap tissue thins, it remains vascularized, limiting best-corrected visual acuity.14
Once neurotrophy advances to perforation, treatment depends largely on size. Small perforations can be glued, while large perforations are generally repaired with patch grafts or penetrating keratoplasties. A penetrating keratoplasty is a full-thickness transplant—the graft type used in most NK cases that lead to perforation. Transplants have a better chance of survival if they can be delayed with temporizing measures.13,27 This allows time for inflammation to subside. Unfortunately, corneal transplants in neurotrophic patients are more likely to fail due to a propensity for continued or even worsening neurotrophy, which leads to chronic, non-healing epithelial defects.5
The prognosis of NK depends largely on severity, duration and comorbidities. Severely neurotrophic corneas are among the most difficult corneal pathologies to effectively manage, as sight is threatened and the potential for recurrence is high. The primary objectives for appropriate management should be to optimize the ocular surface, close epithelial defects and reduce collagenase activity. Careful attention to the many facets of neurotrophy should be given to provide optimal patient care, maintain ocular integrity and ultimately preserve vision.
Dr. Mannen practices at a private optometry and ophthalmology clinic in the greater Ogden, Utah area. She completed her residency at the Walla Walla Veteran Affairs Medical Center and Pacific Cataract and Laser Institute. She is also a fellow of the American Academy of Optometry.
1. Bonini S, Rama P, Olzi D, et al. Neurotrophic keratitis. Eye (Lond). 2003:17(8):989-95. 2. Semeraro F, Forbice E, Romano V, et al. Neurotrophic keratitis. Ophthalmologica. 2014:231(4):191-7. 3. Srinivasan S, Lyall DAM. Clinical Gate. Neurotrophic keratopathy. clinicalgate.com/neurotrophic-keratopathy/. August 3, 2015. 4. Hamrah P, Cruzat A, Dastjerdi MH, et al. Unilateral herpes zoster ophthalmicus results in bilateral corneal nerve alteration: an in vivo confocal microscopy study. Ophthalmology. 2013;120(1):40-7. 5. Chang BH, Groos EB. Neurotrophic keratitis. In: Cornea: Fundamentals, Diagnosis, and Management. Elsevier; 2017:1035-42. 6. Petropoulos IN, Alam U, Fadavi H, et al. Rapid automated diagnosis of diabetic peripheral neuropathy with in vivo corneal confocal microscopy. Invest Ophthalmol Vis Sci. 2014;55(4):2071-8. 7. Okoro CC, Amiebenomo OM, Aruotu N. Medicamentosa keratoconjunctivitis: a case report. Afr Vis Eye Health. 2016;75(1). 8. Sacchetti M, Lambiase A. Diagnosis and management of neurotrophic keratitis. Clin Ophthalmol. 2014;8:571-9. 9. Ralph RA. Tetracyclines and the treatment of corneal stromal ulceration: a review. Cornea. 2000;19(3):274-7. 10. Lambiase A, Rama P, Aloe L, et al. Management of neurotrophic keratopathy. Curr Opin Ophthalmol. 1999;10(4):270-6. 11. Yoon KC, You IC, Im SK, et al. Application of umbilical cord serum eyedrops for the treatment of neurotrophic keratitis. Ophthalmology. 2007;114(9):1637-42. 12. Kaufman SC. Anterior segment complications of herpes zoster ophthalmicus. Ophthalmology. 2008;115(2 Suppl):S24-32. 13. Solomon A, Meller D, Prabhasawat P, et al. Amniotic membrane grafts for nontraumatic corneal perforations, descemetoceles, and deep ulcers. Ophthalmology. 2002;109(4):694-703. 14. Mantelli F, Nardella C, Tiberi E, et al. Congenital corneal anesthesia and neurotrophic keratitis: diagnosis and management. Biomed Res Int. 2015;2015:805876. 15. Portnoy SL, Insler MS, Kaufman HE. Surgical management of corneal ulceration and perforation. Surv Ophthalmol. 1989;34(1):47-58. 16. Shaheen BS, Bakir M, Jain S. Corneal nerves in health and disease. Surv Ophthalmol. 2014;59(3):263-85. 17. Jeng BH, Dupps WJ Jr. Autologous serum 50% eyedrops in the treatment of persistent corneal epithelial defects. Cornea. 2009;28(10):1104-8. 18. Chikama T, Fukuda K, Morishige N, et al. Treatment of neurotrophic keratopathy with substance-P-derived peptide (FGLM) and insulin-like growth factor I. Lancet. 1998;351(9118):1783-4. 19. Yanai R, Nishida T, Chikama T, et al. Potential new modes of treatment of neurotrophic keratopathy. Cornea. 2015;34(Suppl 11):S121-7. 20. Bonini S, Lambiase A, et al. Phase II randomized, double-masked, vehicle-controlled trial of recombinant human nerve growth factor for neurotrophic keratitis. Ophthalmology. 2018;125(9):1332-43. 21. Bonini S, Lambiase A, Rama P, et al. Topical treatment with nerve growth factor for neurotrophic keratitis. Ophthalmology. 2000;107(7):1347-51. 22. Lambiase A, Rama P, Bonini S, et al. Topical treatment with nerve growth factor for corneal neurotrophic ulcers. N Engl J Med. 1998;338(17):1174-80. 23. Koaik M, Baig K. Corneal neurotization. Curr Opin Ophthalmol. 2019;30(4):292-8. 24. Terzis JK, Dryer MM, Bodner BI. Corneal neurotization: a novel solution to neurotrophic keratopathy. Plast Reconstr Surg. 2009;123(1):112-20. 25. Jhanji V, Young AL, Mehta JS, et al. Management of corneal perforation. Surv Ophthalmol. 2011;56(6):522-38. 26. Setlik DE, Seldomridge DL, Adelman RA, et al. The effectiveness of isobutyl cyanoacrylate tissue adhesive for the treatment of corneal perforations. Am J Ophthalmol. 2005;140(5):920-1. 27. Nurözler AB, Salvarli S, Budak K, et al. Results of therapeutic penetrating keratoplasty. Jpn J Ophthalmol. 2004;48(4):368-71. |


