 |  |
Presbyopia is a near-certainty for anyone who lives to reach the age at which the eye’s natural accommodative ability begins to fail. By the time patients are 50 years old, nearly 100% require some type of refractive correction. There is no other condition we manage where the same holds true. Yet only a small percentage of these patients wear contact lenses. We have the opportunity to step in and satisfy this unmet need.
Ophthalmic lens and contact lens technologies present solutions for these patients. Unfortunately, many aren’t aware that there are contact lens options available, as they may not have been given the chance to try them. In the near future, there will also be pharmaceutical options to treat the symptoms of presbyopia. It is critical that we help our patients understand all of their options so that we can improve their outcomes and our standard of care.
Daily Disposables
Historically, daily disposable lenses have been somewhat limited in their parameter availability, making it difficult for some patients to successfully wear them. This should not overshadow their advantages, especially as there has been an expansion in power ranges. These lenses are worn and disposed of on a daily basis, so there is no interaction with cleaning and disinfecting solutions, and patients are guaranteed a clean, fresh lens wearing experience each day. Daily disposables are an ideal option for patients who stand to benefit from contact lenses and prefer part-time wear.
One complaint patients may have about daily disposable lenses is the amount of waste they produce. Fortunately, TerraCycle offers a program for patients to recycle lenses and packaging, and manufacturers like Bausch + Lomb have taken the initiative to encourage recycling.
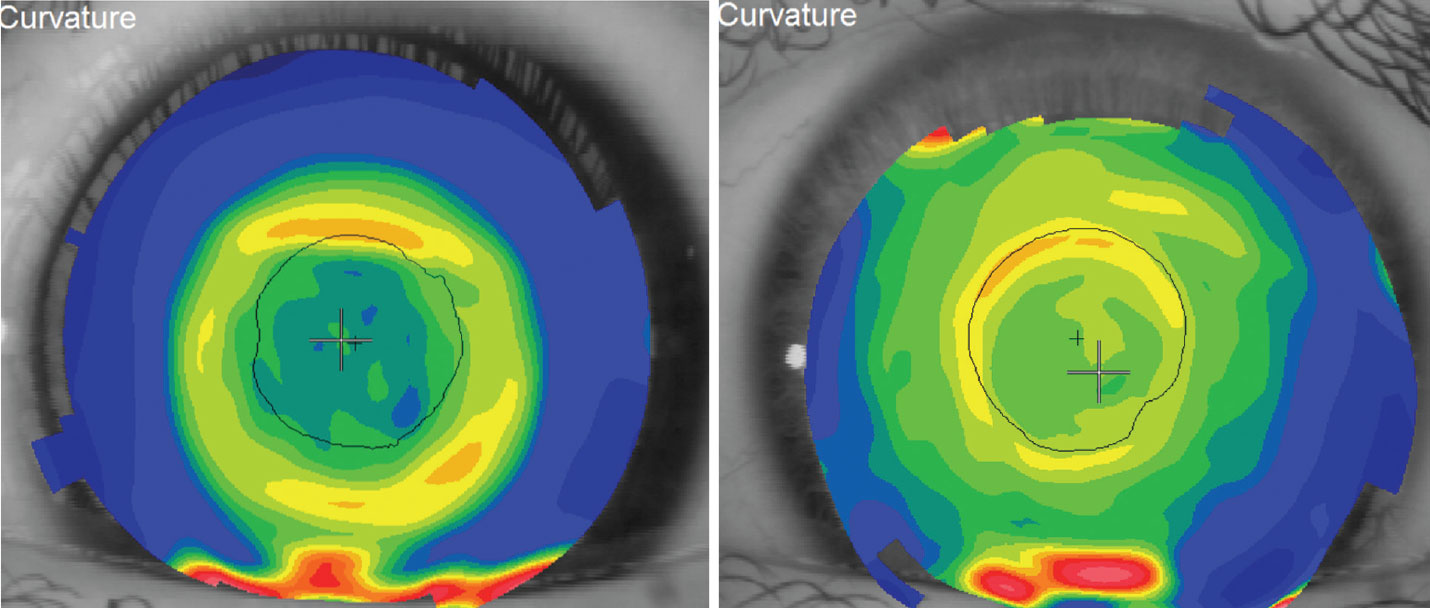 |
| Fig. 1. Topography of a successful orthokeratology patient’s cornea (left). Topography over the surface of a distance-centered multifocal contact lens (right). Click image to enlarge. |
Specialty Lenses
There are currently no daily disposable options available to presbyopic patients who have astigmatic refractive error. Those interested in daily disposables are often limited to best-corrected distance vision in contact lenses and a combination of reading glasses and contact lenses to see at near. Monovision could help reduce the dependency for near glasses over contact lenses.
We have had access to specialty soft contact lens designs for decades. These lenses give patients with multifocal requirements who also need astigmatic refractive correction the opportunity to wear contact lenses. The inherent challenge with these lenses is that they are custom made and come with a wait time for initial access and orders and reorders in the case of modifications.
Presbyopic lenses with astigmatic correction have experienced advances in recent years. There is now a monthly disposable silicone hydrogel lens Ultra Multifocal for Astigmatism (Bausch + Lomb) with toric and multifocal correction. The lens is made of samfilcon A and is 46% water. This lens is unique in that it is available in a diagnostic set to help you avoid much of the wait time in the ordering process.
Other Options
As a profession, optometry has become increasingly aware of line of sight and how it may affect a patient’s visual performance in soft multifocal designs. This can be seen through the advancements the field has made. We now have a contemporary soft multifocal design and a scleral lens design we can customize to offset the optics nasally to correspond with a patient’s line of sight.
Gas permeable (GP) lenses are a viable option for presbyopes, even though they often are thought of as a secondary option for patients who may experience initial lens awareness. GPs provide optical clarity and are intuitively designed to provide distance vision correction in the center of the lens while progressing to the near powers toward more peripheral portions. Translation of the lens in downgaze allows patients to acquire more near power.
Hybrid lenses provide opportunities for presbyopes as well. These lenses have a GP center that is surrounded by a soft lens skirt. This makes initial lens awareness subtler, similarly to soft lenses, and offers comparable optical quality properties to standard GP lenses.
Although much of the conversation surrounding orthokeratology over the last several years has revolved around managing myopia, we certainly can’t overlook our presbyopic patients as potential candidates who are looking for alternatives to glasses or traditional contact lenses. As such, myopic presbyopes are a logical group to consider with this lens technology. Because of the reverse curve in the lens design, the cornea has a prominent steep curve around the pupil. We could look at orthokeratology as having a similar effect as distance-centered, near periphery soft multifocal lenses (Figure 1). As presbyopia progresses, the appropriate next step would seem to be inducing monovision with the lens by under-correcting the level of myopia in the non-dominant eye.
Hyperopic-correcting ortho-K lenses are also now available. As opposed to placing pressure on the central portion of the cornea, they put pressure on more peripheral portions of the cornea, steepening the central cornea and inducing myopic refractive correction. When done over the non-dominant eye, it can have a monovision effect, promoting better near vision.1,2
Pharmaceutical Treatments
There are several pharmaceutical options on the horizon. It is critical that contact lens practitioners understand how to use these new technologies to supplement the contemporary contact lens practice.
In early presbyopes, this may replace the need for multifocal contact lenses, allowing patients to continue with single vision lenses while using drops. Depending on the efficacy of the drop, it may negate the need for multifocal technologies for more advanced presbyopes as well. Some s may still need presbyopic refractive correction in addition to pharmaceutical assistance, although to a lower degree than what would be expected. This could be achieved with lower add-powered multifocals, which are beneficial because there is less of a discrepancy between the distance and near optics within the lens, maximizing the chances of a patient’s success.
There are several presbyopic drops currently under development to be aware of. EV06 1.5% (UNR844-Cl) by Novartis is a lipoic acid choline ester that breaks disulfide bonds, which are thought to harden the lens over time.3,4 By disrupting these bonds, the lens becomes more elastic and regains some functionality. PRX-100 by Presbyopia Therapies, CSF-1 by Orasis Pharmaceuticals and AGN-199201 and AGN-190584 by Allergan are miotic treatments that create a pinhole effect to allow for a greater depth of focus.5-8
Leveraging these technologies to enhance contact lens success will improve the presbyopic experience by giving patients more freedom from spectacle wear.
With current contact lens technologies and the promise of future therapies, we should have no problem helping presbyopic patients achieve clear vision and a comfortable lifestyle and fulfilling a need that has been neglected.
1. Williams BT. Orthokeratology for hyperopia and presbyopia. Contact Lens Spectrum. www.clspectrum.com/issues/2016/august-2016/orthokeratology-for-hyperopia-and-presbyopia. August 1, 2016. Accessed February 7, 2020. 2. Gifford P, Swarbrick HA. Refractive changes from hyperopic orthokeratology monovision in presbyopes. Optom Vis Sci. 2013;90(4):306-13. 3. Pending presbyopia treatments edge closer to disrupting the marketspace. Healio. www.healio.com/ophthalmology/cornea-external-disease/news/print/ocular-surgery-news/%7Ba40b0eb9-cddf-4908-b9bb-9911bef60bf1%7D/pending-presbyopia-treatments-edge-closer-to-disrupting-the-marketspace. March 25, 2019. Accessed February 7, 2020. 4. A study of safety and efficacy of UNR844 chloride (UNR844-Cl) eye drops in subjects with presbyopia. ClinicalTrials.gov. clinicaltrials.gov/ct2/show/NCT03809611. January 18, 2019. Accessed February 7, 2020. 5. Evaluation of the efficacy and safety of PRX-100 in the treatment of early to moderate presbyopia. ClinicalTrials.gov. clinicaltrials.gov/ct2/show/NCT02554396. September 18, 2015. Accessed February 7, 2020. 6. Safety, tolerability and efficacy of PresbiDrops (CSF-1) in presbyopia subjects. ClinicalTrials.gov. clinicaltrials.gov/ct2/show/NCT02965664. November 17, 2016. Accessed February 7, 2020. 7. Orasis Pharmaceuticals. www.orasis-pharma.com/. Accessed February 7, 2020. 8. A safety, efficacy and pharmacokinetic study of AGN-199201 and AGN-190584 in patients with presbyopia. ClinicalTrials.gov. clinicaltrials.gov/ct2/show/NCT02780115. May 23, 2016. Accessed February 7, 2020. |


