The ocular surface is the thin yet complex layer of defense against potentially devastating adverse effects on the refractive ability of the eye. Its tear film is comprised of nutrients, proteins and anti-inflammatory components within a rich mucin-laden adhesive that is kept intact by an even thinner layer of meibum. The successful production of this muco-adhesive layer is dependent on an intact sensory-autonomic reflex, comprised of a healthy corneal sensation and lacrimal gland function. This delicate homeostatic balancing act has no safety net to catch a failing ocular surface, with one exception: us.
Optometrists know firsthand how essential this layer is to maintaining the optical qualities that our patients demand. As innovation created new opportunities to care for patients, modern technology has provided patients with ways to correct their ametropia without the need for spectacles and contact lens materials and solutions have flourished. Such technological advancement has also been witnessed in the area of refractive surgery, which has evolved from its rather primitive origins into a highly precise and predictable protocol for refractive error correction. However, the quality of these results still rests on the continued sustainability of that minuscule layer coating the epithelium. Any imbalance in the delicate system—comprised of a healthy and functioning tear film plus a normal blink reflex with well-positioned eyelid apposition—will ultimately lead to an unhealthy ocular surface.
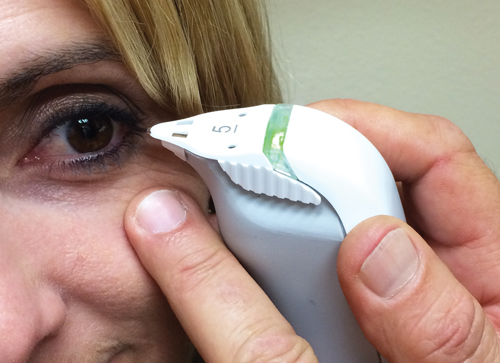 | |
| TearLap can be used to quantify a patient's dry eye. |
The definition of refractive surgery has changed significantly in the two decades since LASIK was approved, blurring the line between refractive and cataract surgeries. The use of smaller corneal incisions during cataract surgery and the ability to incorporate laser into our surgical suite, as well as the increase in lens options (both multifocal and toric) has repositioned the pseudophake under the refractive umbrella. Cataract surgery in particular has been made better with the introduction of the femtosecond laser; however, laser surgery also comes with some complications.
Virtually all laser eye surgery patients experience some degree of dry eye in the first six months after surgery, with symptoms persisting beyond that in nearly 20% of patients.1 Subclinical preexisting dry eye demonstrates the highest risk of long-term postoperative dry eye symptoms, possibly for up to 16 months postoperatively.1 Peak dryness in cataract surgery patients in particular occurs around one month after surgery, and may last longer than three.2 Some practitioners believe that the damage to the corneal nerve plexus during both cataract and refractive procedures leads to neurotrophic dry eye. The incisions used to enter the epithelium, deep stromal nerve changes during ablation, application of suction rings to the mucin-secreting goblets cells and microvilli all play a role in perpetuating inflammatory dry eye.3 The increase in this dryness may also be exacerbated by the necessary use of topical medications, whereby their sequelae is toxic to the epithelium.
Preparation
As partners in our patients’ desire to obtain the best quality of vision, we need to be able to recognize and treat any irregularities in the patient’s ocular surface. Thus, we need to identify these patients by any means necessary to prevent complications. Historically, we have relied on patient complaints or corneal staining to identify these abnormalities; however, these methods can be misleading since the predictive diagnostic value is not consistently high.
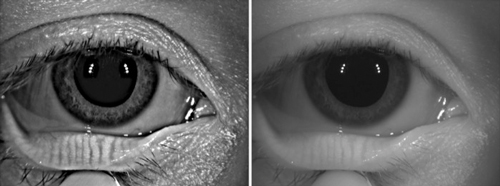 | |
| Oculus meibography. |
The first step toward achieving a successful surgical outcome is to record a thorough medical history. Common preoperative and intraoperative risk factors for dry eye with refractive surgery include the depth of the laser incision, degree of preoperative myopia, female gender, contact lens use, diabetes and eyelid disorders; thus, the evaluation should include an examination of lid anatomy and function, blink rate and tear film composition (including osmolarity). Clinicians should also inquire about patients’ concurrent use of systemic medications that may increase dryness, the frequency with which they use artificial tears and the humidity of their living environment, as these can inhibit recovery following surgery. Note, patients with systemic inflammatory diseases, such as Sjögren’s syndrome and rheumatoid arthritis, are often poor surgical candidates based on their pro-inflammatory status.
Identification
Several procedures can help detect preoperative dry eye in cataract patients. Tear hyperosmolarity, defined as a tear concentration with a greater than average mOsm/L, is a highly accurate marker of dry eye and can also adversely affect cataract outcomes: one study found 21% of patients with hyperosmolarity had a measurement between 0.5D and 1.0D, and 16% had more than 1D of change in keratometry readings.4 The TearLab osmolarity test can be used both to identify dry eye and quantify its severity.
The presence of certain levels of matrix metalloproteinase (MMP) inflammatory mediators can also be used to indicate an abnormal ocular environment. InflammaDry (RPS) is one method of objectively determining the levels of MMP-9, which has been associated with inflammatory dry eye disease.5 Tear analysis can also be used to assess dry eye conditions in pre-operative patients by exposing the tear film and meibomian glands to a light source to highlight any abnormalities. Equally important is an assessment of the number and functional level of lipid-secreting meibomian glands, as well as an evaluation of meibum consistency. This can be done using meibography. In addition to evaluating meibum quality, gland integrity should also be considered. Using a transilluminator to evert the lid and viewing the retroillumination at the slit lamp can be a way to see these glands without a meibography.
Postoperative Evaluation
Because postoperative dry eye can compromise visual acuity and adversely affect an otherwise near-perfect visual outcome, it is important to evaluate the patient’s refractive ability postoperatively to ensure the results are adequate. Whereas in the past we could anticipate patients being within a diopter or two of the intended goal, we now measure success in quarter-diopter steps.
Dry eye can lead to different postoperative complications depending on the procedure. In the case of the KAMRA corneal inlay (AcuFocus), for example, which allows only central light rays to reach the retina through a 1.6mm diameter hole, the presence of dryness on the central aspect of the cornea can distort the final image.
Cyclosporine is a useful treatment for reducing postoperative dry eye in patients with marked preoperative hyperosmolarity or tear film abnormality. Starting your patient on a twice-daily dose of Restasis (Allergan), combined with an artificial tear that is reflective of their lid and osmolarity readings, can foster a more hospitable environment. Typically, the patient should stay on this regimen for a minimum of four to six weeks before measurements are retaken. Macrolide drops or lid scrubs such as HypoChlor (OcuSoft) can be used to reduce bacterial colonization on the lid margin to prevent infection, while warm compresses can help to soften meibomian gland issues. Punctal plugs are also extremely useful in the treatment of dry eye and can be placed before any surgery is performed; however, inflammation should be reduced prior to insertion.
Lastly, nutritional therapy may also play an important role in preoperative treatment of dry eye. Fish oil containing eicosapentaenoic and docosapentaenoic polyunsaturated fatty acids have been shown to reduce inflammation, while omega-3 fatty acids decrease inflammation, increase tear production and may potentiate the regeneration of nerves.6 Directing patients to take high-dose capsules containing omega-3 and omega-6 fatty acids on a daily basis beginning one month before surgery can help maintain the tear film.
Postoperative dry eye treatment should follow the same steps of detection and therapy as in the preoperative period. Note, however, since patients are now dealing with severed nerves and the potential adverse response to their medication, they should stay on any therapy instituted prior to surgery. Restasis drops and artificial tears should be used for a minimum of 90 days postoperatively, with further testing to determine the long-term use. Lid disease should be treated on the same level. Additionally, check for variations in best-corrected visual acuity on every follow-up visit.
Patient expectations are increasing to match advances in ocular surgery; thus, clinical management of the ocular surface before and after arguably becomes as critical to cataract and refractive surgeries as the actual surgical process. Evaluation of patients with diligence toward improving quality of vision and minimizing dry eye symptoms is a responsibility we all share. This is a process that should start from the moment the patient wants to improve their vision through any intervention—be it glasses, contacts or surgery. Yet, one can always stop wearing contacts or change the lenses in glasses; refractive surgical outcomes are not as pliable. So, remember: surgical success starts with a good consultation, diagnosis and unique ocular surface treatments.
Dr. Bloomenstein is the director of optometric services at the Schwartz Laser Eye Center in Scottsdale, Arizona. He is a founding member of the OCRT and a past-president of the Arizona Optometric Association.
1. Bailey, MD, Zadnik K. Outcomes of LASIK for myopia with FDA-approved lasers. Cornea. 2007;26(3):246-254.
2. Li XM, Hu L, Hu J, Wang W. Investigation of dry eye disease and analysis of the pathogen factors n patients after cataract surgery. Cornea. 007 Oct;26(9 Suppl 1):S16-20.
3. Oh T, Jung Y, Chand D, et al. Changes in the tear film and ocular surface after cataract surgery. Jpn J Ophthalmol. 2012 Mar;56(2):113-8.
4. Epitropoulos AT, Matossian C, Berdy GJ, et al. Effect of tear osmolarity on repeatability of keratometry for cararact surgery planning. J Cataract Refract Surg. 2015 Aug;41(8):1672-7.
5. Kaufman HE. The practical detection of MMP-9 diagnoses ocular surface disease and may help prevent its complications. Cornea. 2013 Feb;32(2):211-6.
6. Root M, Collier SR, Zwetsloot KA, et al. A randomized trial of fish oil omega-3 fatty acids on arterial health, inflammation and metabolic syndrome in a young healthy population. Nutrition Journal 2013 Apr 8;8:40.


