Have you noticed corneal disease patients who present with a rise in intraocular pressure, developing cataracts and secondary infections? As you may have guessed, these are all potential adverse effects of corticosteroid use in the treatment and management of corneal disease. However, in my experience, I more frequently see complications due to the withholding of a corticosteroid in corneal disease when it should have been prescribed.
Such undue caution can result in corneal neovascularization, prolonged infiltrative keratitis, uncontrolled uveitis, persistent patient symptoms and even corneal scarring. In a number of these cases, the patient was left with only one option—corneal transplant. That said, I add the caveat that practitioners must always be cautious in the use of corticosteroids, and instruct patients to use them appropriately so as to minimize any risks.
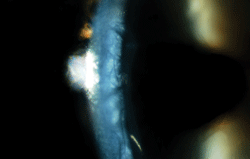
Corneal edema.
Aside from prescriptions related to surgical care, optometry writes more prescriptions for certain steroids, such as Lotemax (loteprednol 0.5%, Bausch + Lomb) than any other medical profession. So, while we are comfortable with the use of corticosteroids, it can be helpful to have a refresher course on the do’s and don’ts of proper use.
Corticosteroids
When incorporating steroids into your treatment plan, there are three potential concerns: IOP elevation (which could lead to glaucoma), secondary infections due to the immunosuppressive nature of corticosteroids and the formation of posterior subcapsular cataracts.
Newer versions of these agents—ester-based steroids—have significantly reduced complication rates. One recent multicenter study comparing dexamethasone to ketone-free loteprednol found half as many cases of significant IOP elevation with loteprednol.1 These findings were comparable to previous analyses.2 Ketones can form a Schiff base, which is a precursor to cataract development; the absence of a ketone in loteprednol precludes this from occurring.3 However, decreasing the risk of IOP elevation with an ester-based steroid does not eliminate the risk of significant IOP elevation––especially with long-term use.4
Don’ts
• Don’t write refills. This is perhaps more of a personal preference, but I strongly recommend that you do not allow refills––especially on the first prescription for a corticosteroid. Often, the comfort perceived by a patient using topical steroids can lead them to continue the therapy long-term.
If a patient is prescribed multiple refills, they may no longer experience any symptoms and choose not to return to the office, without awareness that their intraocular pressure may be elevated. If you do not write a refill, the patient is forced to return to your office for an IOP check before he or she can receive another prescription.
Similarly, a compromised cornea might preclude the use of corticosteroids, such as in the presence of an abrasion. Once the epithelium is breached, there is an increased risk of potential infection. Suppressing the immune system with corticosteroids in an already present trauma situation, where the epithelium has been debrided, could lead to a potential secondary infection.
• Don’t forget the well-known infectious keratitis contraindications. This includes the presence of epithelial herpes simplex keratitis (whether it be dendritic, geographic, marginal ulcer or any epithelial form). Simply put, patients diagnosed with HSV keratitis in the presence of corneal staining should not be prescribed corticosteroids. Other infectious conditions that could worsen under the presence of a corticosteroid include fungal keratitis, mycobacterium and even bacterial keratitis (especially in the early stages).
The Steroids for Corneal Ulcers Trial (SCUT) found that use of corticosteroids in addition to antibiotics for the treatment of bacterial ulcers did not help or hinder healing or complication rates.5 In fact, more severe bacterial keratitis patients in the SCUT appeared to have a better outcome if corticosteroids were used after the second day and throughout the long-term follow-up.6
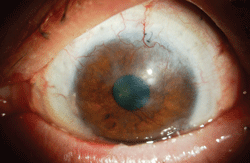
Corneal neovascularization.
It is best to avoid steroids during the early acute phase of a bacterial keratitis; typically, this would mean the first two days for gram-positive bacterial infection and possibly 72 hours for gram-negative rods, such as Pseudomonas aeruginosa.
I would also advise against using corticosteroids when treating a bacterial corneal ulcer, unless a 48- to 72-hour window (depending on the cultured pathogen) has passed and improvement is noted in the clinical picture.
Positive signs include significant epithelialization progress and a pathogen identified via culture that is shown to be susceptible to the drugs being applied. If all of these are met, corticosteroids could indeed decrease the level of inflammation and prevent corneal scarring in a bacterial keratitis.
• Don’t forget to take a detailed patient history. Avoid corticosteroid use on a compromised cornea in patients with uncontrolled systemic diseases (such as arthritis or lupus) when either an infiltrate or corneal thinning is noted. Unless the systemic disease control is well noted, a corticosteroid could lead to increased breakdown of the epithelium because the patient’s systemic disease is causing further corneal damage.
In other words, a corticosteroid might be contraindicated in a patient presenting with rheumatoid arthritis and a corneal melt, which is an indication of poorly controlled rheumatoid arthritis. A better choice of treatment would be systemic immunosuppression via a rheumatologist.
• Don’t mix steroids and contact lenses. There is some debate as to whether a corticosteroid should be applied on top of a contact lens. I believe that the absorption of medication into a contact lens can alter the effects of the drug on the cornea, due to increased contact time (because the lens acts as a drug reservoir). This could lead to prolonged steroid exposure and potentially increase the individual’s risk of complications.
In the giant papillary conjunctivitis trials, patients were administered loteprednol drops on top of a contact lens.7 Even though there were no corneal complications (e.g., infection), the results showed that these patients had a significantly higher incidence of IOP rise vs. studies where a contact lens was not involved. Therefore, it can be assumed that the safety related to IOP rise of even an ester-based corticosteroid is somewhat lost if the drug resides within the contact lens throughout the day. It should also be mentioned that there were no secondary infections reported in any of the patients who received corticosteroid drops directly on the contact lens during clinical trial.7
Dos
• Do use corticosteroids in a timely manner. When a corticosteroid is warranted, withholding it could lead to even greater complications, such as scarring or neovascularization. We have very few medications on the market that are angiotensive—i.e., can prevent corneal vascularization as well as give comprehensive inflammation suppression. For that reason, if corneal neovascularization is present and encroaching the visual axis, it is
critical that aggressive corticosteroid therapy be used.
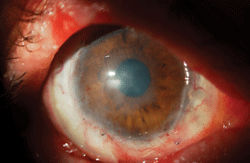
Corneal scarring.
Likewise, infiltrative or inflammatory corneal events require the use of aggressive corticosteroids. However, if a contact lens peripheral infiltrate is noted because the cornea is compromised, it would be more prudent for a practitioner to use a steroid/antibiotic combination agent as opposed to simply pure corticosteroids. Inflammatory conditions tend to be closer to the limbus and often have epithelial defects or staining that is much smaller than the actual infiltrate’s size.
• Do look for critical signs. Always be cautious of the potential signs of an infectious keratitis in the presence of an infiltrate. This typically can be found in patients with significant symptoms, such as acute onset pain and photophobia, a very red eye 360° in the conjunctiva, decreased vision and the presence of discharge.
The critical signs of a bacterial keratitis include lid edema, an epithelial defect over an infiltrate with significant staining and an anterior chamber reaction. If the signs are more indicative of a chronic inflammatory event, use steroids readily and aggressively via a combination steroid/antibiotic drop to help the patient with pain and inflammation.
• Do pick the right delivery method. Because we have so many options and delivery forms of corticosteroids, ranging from suspensions to emulsions to gels and ointments, it is wise to become familiar with all of the different options and how to use them when the appropriate patient presents.
For example, the addition of an overnight steroid ung to daily drops can dramatically improve the level of inflammation when treating iritis. An overnight corticosteroid ointment can also help patients with morning symptoms and/or significant meibomian gland dysfunction.
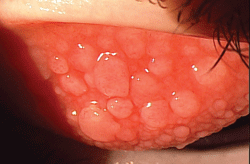
Severe vernal keratoconjunctivitis.
Since resuspension requires 20-30 bottle shakes, try to select medications that do not require resuspension. Three corticosteroid formulations on the market today—Lotemax gel, Lotemax ointment and Durezol (difluprednate 0.05%, Alcon)—report complete uniformity in each drop.
This is critical when treating significant inflammation where an inappropriate amount of drug can result in iritis, uveitis or non-infectious infiltrative keratitis.
Prescribing steroids is essential to successful clinical practice and good patient outcomes. When a patient with a compromised cornea presents, doctors must use caution to determine if corticosteroids could cause harm. Clinicians must look for key signs—everything from infectious keratitis to epithelial defects and even active systemic disease—to determine if a corticosteroid is contraindicated. If not contraindicated, aggressive steroid therapy is warranted and may prevent serious complications.
Dr. Karpecki works in the corneal services and heads the clinical research department at the Koffler Vision Group in Lexington, Ky.
1. Chen M, Gong L, Sun X. A multicenter, randomized, parallel-group, clinical trial comparing the safety and efficacy of loteprednol etabonate 0.5%/tobramycin 0.3% with dexamethasone 0.1%/tobramycin 0.3% in the treatment of Chinese patients with blepharokeratoconjunctivitis. Curr Med Res Opin. 2012 Mar;28(3):385-94.
2. Holland EJ, Bartlett JD, Paterno MR. et al. Effects of loteprednol/tobramycin versus dexamethasone/tobramycin on intraocular pressure in healthy volunteers. Cornea. 2008 Jan;27(1):50-5.
3. Comstock TL, Decory HH. Advances in corticosteroid therapy for ocular inflammation: loteprednol etabonate. Int J Inflam. 2012. [Epub 2012 Mar 28]
4. Raipal RK, Digby D, D’Aversa G. Intraocular pressure elevations with loteprednol etabonate: a retrospective chart review. J Ocul Pharmacol Ther. 2011 Jun;27(3):305-8.
5. Srinivasan M, Mascarenhas J, Rajaraman R et al. Corticosteroids for bacterial keratitis: the Steroids for Corneal Ulcers Trial (SCUT). Arch Ophthalmol. 2012 Feb;130(2):143-50.
6. McClintic SM, Srinivasan M, Mascarenhas J, et al. Improvement in corneal scarring following bacterial keratitis. Eye (Lond). 2013 Mar;27(3):443-6.
7. Bartlett JD, Howes JF, Ghormley NR. Safety and efficacy of loteprednol etabonate for treatment of papillae in contact lens-associated giant papillary conjunctivitis. Curr Eye Res. 1993 Apr;12(4):313-21.


