The evolution of therapeutic management
of ocular disease by eye care practitioners has placed an ever-growing
number of medications at our disposal. One underutilized and
underappreciated class of medications in eye care is non-steroidal
anti-inflammatory drugs (NSAIDs).
Commonly
prescribed to reduce pain and inflammation in a variety of tissues,
NSAIDs are among the most widely used medications in the world because
of their demonstrated efficacy in reducing pain and inflammation.1
Though available in both topical and systemic formulations, topically
applied NSAIDs are more commonly used in the prevention and management
of non-infectious ocular inflammation and cystoid macular edema
following cataract surgery.
These drugs are also used in the management of ocular pain following corneal procedures and in the treatment of allergic conjunctivitis.2
Mechanism of Action
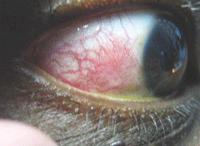
Episcleritis is inflammation of the episclera that occurs
without an infection. It is a common condition that is usually mild.
Treatment with topical anti-inflammatories may relieve the symptoms
faster.

A
corneal abrasion is probably the most common eye injury. The pain may
be severe and should be treated with non-steroidal anti-inflammatory
drops.
In
the inflammation pathway, NSAIDs specifically inhibit the action of
cyclo-oxygenase, an enzyme vital to prostaglandin synthesis.3 Animal
studies have shown that trauma to the anterior segment of the
eye—especially the iris—increases prostaglandin synthesis and may
contribute to the development of ocular inflammation.4 NSAIDs inhibit
prostaglandin synthesis, which makes them very useful in controlling
pain and inflammation without adversely impacting the immune system.
The NSAIDs’ action takes place further down the arachidonic acid
pathway than steroids.
What does that mean clinically? The drugs
are not as effective as steroids in reducing inflammation, but are
still quite potent and lack the side effects seen in steroid usage.2
As a group, though, NSAIDs are excellent analgesics and are even more effective than intramuscular morphine for acute pain.5 The
historical development of NSAIDs is closely related to the historical
development of aspirin.2 In fact, topical NSAIDs are often referred to
as “aspirin for the eye.” The drugs are very useful in the management
of ocular pain following cataract surgery and corneal procedures.
There are a number of NSAIDs approved for topical ophthalmic use. These include:
• Nevanac (nepafenac 0.1%, Alcon).
• Acuvail (ketorolac tromethamine 0.4%, Allergan).
• Acular (ketorolac tromethamine 0.5%, Allergan).
• Voltaren (diclofenac sodium 0.1% , CIBA Vision).
• Xibrom (bromfenac 0.09%, ISTA Pharmaceuticals).
• Profenal, (suprofen 1%, Alcon).
• Ocufen (flurbiprofen 0.3%, Allergan).
Topical NSAIDs and Analgesia
NSAIDs
are FDA-approved for the reduction of pain following refractive
surgery. Patients treated postoperatively with NSAIDs reported
significantly greater pain relief, less pain intensity, fewer symptoms
of ocular discomfort and less sleep discomfort than those who were not
treated with NSAIDs postoperatively.7 Also, many doctors have found it
beneficial to use topically applied NSAIDs in reducing pain after a
corneal abrasion, but effectiveness seems to be greater if the drugs
are administered preoperatively. In fact, studies have shown that this
approach in refractive surgery has the potential to more effectively
inhibit pain.6
Postoperative pain has been a significant
complication of photorefractive keratectomy (PRK), and using bandage
contact lenses, cycloplegia and oral narcotics alone yielded limited
success. Much of this pain and discomfort is modulated once the cornea
re-epithelializes.
Indeed, hyperesthesia may occur following
excimer laser ablation.8 The immediate postoperative use of 0.1%
diclofenac in combination with topical fluorometholone 0.1% and a
bandage contact lens significantly reduced pain, discomfort and
photophobia following PRK in several studies.9,10
These studies
also showed that diclofenac did not interfere with wound healing. One
study reported that diclofenac significantly decreases corneal
sensitivity in the human eye.11 While the mechanism by which this NSAID
decreases corneal sensitivity isn’t known, this finding explains the
pain relief patients experience after receiving this drug. Limited-dose
administration in a compromised cornea is recommended. NSAIDs should be
administered three to four times a day while the patient experiences
pain, usually two to three days postoperatively. They should be avoided
in patients with severe dry eye, as most of the reported cases of
corneal complications with NSAID use occurred in patients whose tear
production was insufficient (and who were concurrently using topical
steroids).12
What are some other surgical uses of NSAIDs that have
proved appropriate? Instillation of NSAIDs for three days prior to and
one day following cataract surgery can prevent ocular pain and
discomfort in the first 24 hours after surgery.13 More importantly,
NSAID use has been shown to significantly lessen the risk and severity
of cystoid macular edema, which can occur after cataract surgery.14-16
Many
surgeons now use them routinely for three to four weeks postoperatively
to reduce the chance of CME.While steroids offer an effective treatment
for CME, NSAIDs are not associated with pressure spikes or other ocular
side-effects.
While significant corneal abrasions that are not
associated with contact lens use generally resolve spontaneously
without complications, they may cause debilitating pain during the
first 24 to 48 hours after the injury. And, this pain is what drives
the patient to present for treatment. Several studies have been
undertaken to evaluate the role of NSAIDs in the management of corneal
abrasions not due to infections, contact lens trauma or foreign body
removal. Patients receiving a topical NSAID required significantly
fewer additional oral analgesics and resumed normal activities
sooner.17,18 In patients with acute corneal abrasions, ophthalmic
NSAIDs reduce pain without a delay in healing; NSAID use can make the
difference for those individuals unable to miss work because of ocular
discomfort.19
Many forms of uveitis require prolonged steroid
therapy to control inflammation. Therapy may need to be escalated to
include oral steroids or periorbital injections in recalcitrant cases,
with the risk of local toxicity and adverse events increasing
substantially. NSAIDs are a safer alternative treatment in certain
forms of uveitis.20 Painful ocular conditions, such as episcleritis and
corneal limbal ulcers, may benefit not only from the steroid-sparing
effects of NSAIDs but from their analgesic properties as well.21
Oral NSAIDs and Pain Relief
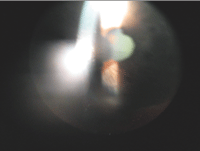
Anterior uveitis presents with anterior chamber cell
and flare, and chronic uveitis often shows posterior synechiae and keratitic precipitates, as seen here.
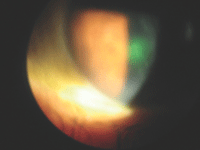
Recurrent
corneal erosion often presents following a previous corneal abrasion.
The epithelium is loose around the defect and can be recalcitrant to
treatment.
There
are hundreds of proprietary analgesics in the marketplace, and, of
course, all of their manufacturers claim their effectiveness. Many
physicians and patients are confused as to which analgesic works best
for their pain, and often the choice is based on personal experience
rather than evidence.22,23 Evidence in favor of NSAIDs is generally
overwhelming when the test drug is compared to placebo in acute or
chronic pain conditions.24 However, there is a controversy about the
relative efficacy of NSAIDs when compared with each other.
In the
past, some authors have stated that there is little difference in the
analgesic efficacy between the different types of NSAIDs.5 But, recent
evidence has shown that individual NSAIDs do differ in their analgesic
effect. The Oxford League Table has been suggested as a tool for
assessing the relative efficacy of analgesics—it assigns each analgesic
a number with which to grade its efficacy.25 Analgesic efficacy is
expressed as the “number needed to treat” (NNT), the number of patients
who need to receive the active drug to achieve at least 50% relief of
pain vs. placebo over a four- to six-hour treatment period. The most
effective drugs have a low NNT: just over two, which means that for
every two patients who receive the drug, one patient will experience at
least 50% relief thanks to of the treatment. Many doses of NSAIDs have
NNT values between two and three; ibuprofen 800mg, ketorolac 20mg and
diclofenac 100mg have NNT values of less than two.25 Ibuprofen 800mg is
at the top of the Oxford League Table with a NNT of 1.6 and with 100%
of patients achieving at least 50% pain relief. By comparison, other
analgesics, such as aspirin 600mg (NNT: 4.4) and acetaminophen 100mg
(NNT: 3.8) are significantly less effective. While NSAIDs are more
effective than acetaminophen, acetaminophen has a better safety profile
than NSAIDs.26
But, oral NSAIDs offer an additional benefit: They
are not controlled substances, which allows those eye care
practitioners without DEA privileges the ability to provide pain
control to their patients.
Risks of Topical NSAID Therapy
While
topical ocular NSAIDs show excellent absorption and penetration into
ocular tissues and display dramatic results, they may have some adverse
effects. Frequently reported adverse reactions of NSAIDs are local in
nature and include transient burning, stinging and ocular irritation
upon instillation.18 Hypersensitivity reactions, including itching,
redness and photosensitivity, may also occur.18
Although
manufacturers have used various methods and formulations to minimize
potential discomfort, the use of NSAIDs without concomitant steroids
following PRK has been associated with the development of corneal
infiltrates.9 More serious but less common local corneal complications
include superficial punctate keratitis, epithelial defects, corneal
melting and delayed wound healing.27,28 There have been rare reports of
corneal infiltrates and corneal perforation.29
As a prodrug,
nepafenac may offer an additional safety factor, since less active drug
would be present on the cornea. Studies have shown that nepafenac
inhibits retinal inflammation and exhibits superior corneal penetration
vs. other NSAIDs.30,31 Patients with a history of herpes simplex
keratitis should be monitored while using NSAIDs. And, there are no
adequate studies in pregnant women, so topical NSAIDs should be avoided
in these patients.
Systemic adverse reactions, while rare, have been
reported in the literature. These include exacerbation of bronchial
asthma due to the systemic absorption of topical NSAIDs, as well as
gastrointestinal irritation and ulceration, inhibition of platelet
function and renal disease.32,33 The high-risk group includes geriatric
patients; users of alcohol, tobacco and antacids; and those with a
history of systemic steroid or anticoagulant therapy.34 While the
systemic absorption of topically applied medication is generally
considered minimal, idiosyncratic drug reactions may occur. It is wise
to obtain a complete medical and drug history to see if there are any
potential adverse reactions before starting topical NSAID therapy.
Safe, Effective Treatment
The
development of a safe alternative to steroids in the treatment of
ocular inflammation is a significant advancement in ocular
therapeutics. Unlike topical steroids, topical NSAID therapy rarely
results in serious local or systemic complications. Topical NSAIDs also
provide an analgesic effect and reduce the need for additional oral
analgesics. This class of drugs provides yet another weapon in our
battle against ocular disease.
Dr. Bowling is center director
at Vision America, a surgical comanagement center in Gadsden, Ala. He
is also a diplomate in the Primary Care Section of the American Academy
of Optometry.
Dr. Russell is in group practice with the Marietta Eye
Clinic in Georgia. He is a diplomate in the Section on Cornea, Contact
Lenses and Refractive Technologies of the American Academy of Optometry.
1. Laine L. Approaches to nonsteroidal
anti-inflammatory drug use in the high-risk patient. Gastroenterology.
2001 Feb;120(3):594-606.
2. Colin J. The role of NSAIDs in the management of postoperative ophthalmic inflammation. Drugs. 2007;67(9):1291-308.
3. Janpol LM. Pharmacologic therapy of aphakic cystoid macular edema. Ophthalmology. 1985 Jun;92(6):807-10.
4.
Keulen-DeVos HCJ, Van Rij G, Renardel DeLavalette JCG, et al. Effect of
indomethacin in preventing surgically induced miosis. Br J Ophthalmol.
1983 Feb;67(2):94-6.
5. Ong CKS, Lirk P, Tan CH, et al. An
evidence-based update on nonsteroidal anti-inflammatory drugs. Clin Med
Res. 2007 Mar;5(1):19-34.
6. Colin J, Paquette B. Comparison of the
analgesic efficacy of nepafenac ophthalmic suspension compared to
diclofenac ophthalmic solution for ocular pain and photophobia after
excimer laser surgery: a phase 2 randomized, double masked trial. Clin
Ther. 2006 Apr;28(4):527-36.
7. Yee RN, The Ketorolac Radial
Keratotomy Study Group. Analgesic efficacy and safety of nonpreserved
ketorolac tromethamine ophthalmic solution following radial keratotomy.
Am J Ophthalmol. 1998 Apr;125(4):472-80.
8. Ishikawa T, Cerro M,
Liang FQ, et al. Hypersensitivity following excimer laser ablation
through the corneal epithelium. Refract Corneal Surg. 1992
Nov-Dec;8(6):466-74.
9. Sher NA, Frantz JM, Talley A, et al. Topical
diclofenac in the treatment of ocular pain after excimer
photorefractive keratectomy. Refract Corneal Surg. 1993
Nov-Dec;9(6):425-36.
10. Eiferman RA, Hoffman RS, Sher NA. Topical
diclofenac reduces pain following photorefractive keratectomy. Arch
Ophthalmol. 1993 Aug;111(8):1022.
11. Szerengi K, Sorken K, Garbus JJ, et al. Decrease in normal human corneal sensitivity with topical diclofenac sodium. Am J
Ophthalmol. 1994 Sep 15;118(3):312-5.
12. Donzis PB, Mondino BJ. Management of noninfectious corneal ulcers. Surv Ophthalmol. 1987 Sep-Oct;32(2):94-110.
13.
Price MO, Price FW. Efficacy of topical ketorolac tromethamine 0.4% for
control of pain or discomfort associated with cataract surgery. Curr
Med Res Opin. 2004 Dec;20(12):2015-9.
14. Flach AJ, Stegman RC,
Graham J, Kruger LP. Prophylaxis of aphakic cystoid macular edema
without corticosteroids. A paired-comparison, placebo-controlled
double-masked study. Ophthalmology. 1990 Oct;97(10):1253-8.
15.
Flach AJ, Dolan BJ, Irvine AR. Effectiveness of ketorolac tromethamine
0.5% ophthalmic solution for chronic aphakic and pseudophakic cystoid
macular edema. Am J Ophthalmol. 1987 Apr 15;103(4):479-86.
16. Rho
DS. Treatment of acute pseudophakic cystoid macular edema: diclofenac
versus ketorolac. J Cataract Refract Surg. 2003 Dec;29(12):2378-84.
17.
Goyal R, Shankar J, Fone DL, et al. Randomized controlled trial of
ketorolac in the management of corneal abrasions. Acta Ophthalmol
Scand. 2001 Apr;79(2):177-9.
18. Kaiser PK, Pineda R, Corneal
Abrasion Patching Study Group. A study of topical nonsteroidal
anti-inflammatory drops and no pressure patching in the treatment of
corneal abrasions. Ophthalmology. 1997 Aug;104(8):1353-9.
19. Weaver
CS, Terrell KM. evidence-based emergency medicine. Update: do
ophthalmic nonsteroidal anti-inflammatory drugs reduce the pain
associated with simple corneal abrasion without delaying healing? Ann
Emerg Med. 2003 Jan;41(1):134-40.
20. Samiy N, Foster CS. The role
of non-steroidal anti-inflammatory drugs in ocular inflammation. Int
Ophthalmol Clin. 1996 Winter;36(1):195-206.
21. Koay P. The emerging
role of topical non-steroidal anti-inflammatory agents in
ophthalmology. Curr Med Res Opin. 2005 Jul;21(7):1131-7.
22. Connolly TP. Cyclooxygenase-2 inhibitors in gynecologic practice. Clin Med Res. 2003 Apr;1(2):105-10.
23. Ong CK, Seymour RA. Pathogenesis of postoperative oral surgical pain. 2003 Winter;50(1):5-17.
24. McQuay HJ, Edwards JE, Moore RA. Evaluating analgesia: the challenges. Am J Ther. 2002 May-Jun;9(3):179-87.
25.
Oxford League Table of Analgesics in Acute Pain. Available at:
www.medicine.ox.ac.uk/bandolier/Extraforbando/APain.pdf. (Accessed
February 2010).
26. Ong CKS, Lirk P, Tan CH, et al. An
evidence-based update on nonsteroidal anti-inflammatory drugs. Clin Med
Res 2007;5(1):19-34.
27. Gills JP. Voltaren associated with medication keratitis. J Cataract Refract Surg. 1994 Jan;20(1):110.
28.
Shimazaki J, Saito H, Yang HY, et al. Persistent epithelial defect
following penetrating keratoplasty: an adverse effect of diclofenac eye
drops. Cornea. 1995 Nov;14(6):623-7.
29. Guidera AC, Luchs JI, Udell
IJ. Keratitis, ulceration and perforation associated with topical
non-steroidal ant-inflammatory drugs. Ophthalmology. 2001
May;108(5):936-44.
30. Ke TL, Graff G, Spellman JM, Yanni JM.
Nepafenac, a unique nonsteroidal prodrug with potential utility in the
treatment of trauma-induced ocular inflammation: In vitro bioactivation
and permeation of external ocular barriers. Inflammat. 2000
Aug;24(4):371-84.
31. Kapin MA, Yanni JM, Brady MT, et al.
Inflammation-mediated retinal edema in the rabbit is inhibited y
topical nepafenac. Inflammation. 2003 Oct;27(5):281-91.
32. Sharir M. Exacerbation of asthma by topical diclofenac. Arch Ophthalmol. 1997 Feb;115(2):294-5.
33.
Campbell WB, Halushka PV. Lipid derived autocoids. In: Hardman JG,
Limbird LE, eds. Goodman and Gilman’s The Pharmacologic Basis of
Therapeutics, 9th ed. New York: McGraw-Hill. 1990:601-33.
34. Estes
LL, Fuhs DW, Heaton AH,et al. Gastric ulcer perforation associated with
the use of injectable ketorolac. Ann Ann Pharmacother. 1993
Jan;27(1):42-3.


