Those of us with sufficient experience in contact lens practice recall that papillary conjunctivitis was an all-too-common side effect of lens wear in the early years of the modality. Such were the limitations of the first-generation materials, which allowed excessive protein build-up on the lenses to trigger an inflammatory response. “Planned replacement” lenses were heralded for their ability to reduce the incidence of contact lens-induced papillary conjunctivitis (CLPC)—and indeed they did. Yet it persists. Let’s try to understand why, and what can be done about it.
What is CLPC?
The chronic inflammatory condition we know as giant papillary conjunctivitis (GPC) predates contact lens wear, as ocular prostheses and exposed sutures have long been known to cause the condition through mechanical irritation of the ocular surfaces.1 Constant use of soft or gas-permeable (GP) contact lenses can increase the incidence of it, especially in individuals already experiencing other allergy episodes. For greater specificity, the term CLPC is used to describe cases in which contact lens wear is the primary culprit.
CLPC is prevalent in approximately 20% of hydrogel lens and 5% of RGP lens wearers.2
CLPC is not a true form of allergic conjunctivitis, but rather a non-IgE mediated inflammation of external ocular surfaces. As such, it can exist with other forms of ocular allergy.1 There is no increase in IgE or histamine in the tears of CLPC patients.3 CLPC symptoms can increase during allergy and dry eye season.
Case in point: A 17-year-old male presents complaining of red, itchy eyes and contact lens intolerance during the past month. He has a four-year history of successful daily wear of contact lenses. He has been using an OTC medication without relief. On exam, a papillary reaction was noted on the superior palpebral conjunctiva bilaterally (figure 1).
Signs and Symptoms
Patients with CLPC complain of itching, foreign body sensation, decreased contact lens wearing time, blurry vision due to lens decentration, and a white, ropy discharge. These patients may report replacing their contact lenses more frequently than they have in the past. Note: Always suspect CLPC when a patient presents complaining of recent-onset lens discomfort in a lens that has previously been worn comfortably.
Frequent use of contact lenses causes repetitive physical trauma to the upper tarsal conjunctiva, which produces a papillary hypertrophy—often seen as abnormally large “cobblestone” papillae readily apparent on lid eversion.
The appearance and location of the papillary reaction can vary depending on the type of contact lens worn.4 In hydrogel and GP lens wearers, the CLPC tends to be bilateral and uniform across the tarsal plate, while the papillae tend to be more asymmetric in silicone hydrogel lens wearers.
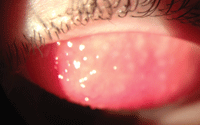
1. Papillary reaction in CLPC.
Remember, to see the papillae you have to evert the lids. Any contact lens patient who complains of discomfort or irritation with contact lens wear, or ptosis, should have their lids everted to examine for CLPC. Rigid lens-related GPC has a localized response, with raised fibrotic white apices generally found closer to the lash region.
The frequency of the patient’s soft contact lens replacement schedule is a major factor in the development of CLPC. A retrospective study of 47 soft contact lens patients showed that patients replacing their lenses at four weeks or longer had a 36% incidence of CLPC, while patients replacing their lenses at less than four weeks had only a 4% to 5% incidence of CLPC. No patients wearing two-week replacement or daily disposable contact lenses developed CLPC in that study.5
CLPC stems from debris buildup on the lens surface, which leads to inflammation that produces even more lens deposits, thus continuing the cycle.6 Complications of CLPC come from immune reactions to the accumulated residue deposited on the contact lens, which may contain cellular debris, contact lens solution preservative deposits and mucus.7
Treatment
Treatment of CLPC is two-fold. The first step is to decrease or eliminate the antigenic load and mechanical irritation from the contact lens itself. The next step is to balance the inflammatory response.
Decreasing contact lens coatings entails improving lens cleaning, decreasing wear time, shortening the lens replacement interval or changing the lens material or design.7 Medications that can temper the inflammation include topical steroids, topical non-steroidal anti-inflammatory agents and mast-cell stabilizers.4
I always strongly recommend the patient temporarily discontinue contact lens wear for several weeks to decrease the antigenic load and remove the mechanical stimulus that produced the CLPC in the first place. However, convincing patients to discontinue contact lens wear, even for a relatively short period of time, can prove difficult.
If the patient has a mild to moderate case of CLPC, I prescribe a “soft” steroid—usually Lotemax (loteprednol 0.5%, Bausch + Lomb) two to four times daily, depending on the severity of the symptoms and CLPC presentation. It should be instilled 10 minutes prior to contact lens insertion and twice in the evening after contact lens removal for two weeks.8
I also recommend a combination antihistamine/mast cell stabilizer—usually Pataday (olopatadine 0.2%, Alcon) one drop in each eye BID. A recent Japanese study confirmed that this agent was found to be generally safe, well tolerated and effective when dispensed in this BID dosage.9 Keep in mind that while olopatadine 0.2% drops are designed and labeled for once-daily dosing, olopatadine and other members of its class—e.g., Elestat (epinastine 0.5%, Allergan), Zaditor (ketotifen, Novartis), Alaway (ketotifen, Bausch + Lomb) and Optivar (azelastine 0.05%, Meda Pharmaceuticals)—are very safe and can be used more often if necessary.10 The combination of olopatadine and a soft steroid was found effective in treating CLPC.11
Have the patient return for follow-up in two weeks to monitor papillary reaction and intraocular pressure. Depending on the presentation, I may taper the steroid to twice daily for another two weeks or discontinue use altogether.
Once the steroid is discontinued, the patient remains on the antihistamine/mast-cell stabilizer long-term. Mast cell stabilizers have been shown to be effective for CLPC, with one study reporting a 70% success rate in patients who had experienced a return of symptoms.12
Finally, the patient’s contact lens and disinfection system should be addressed. If the patient is averse to temporarily discontinuing contact lens wear, then advise switching to a more frequent replacement schedule, daily disposable lenses being the best option. Increasing replacement frequency or transitioning to a daily disposable lens has a success rate of more than 90%, according to one study.13 Also consider recommending a hydrogen peroxide disinfection care system, which is known to be an effective protein remover. Lastly, advise the patient to reduce their contact lens wearing time during periods of allergic activity.
Once diagnosed, patients need to be educated about the chronic nature of CLPC and its symptoms, and reminded to present for care should symptoms reappear. The case mentioned above resolved nicely by temporarily discontinuing the patient’s contact lens wear and prescribing a short course of topical steroids and combination mast cell stabilizer.
Dr. Bowling runs a solo private optometric practice in Gadsden, Ala. He is also a diplomate in the Primary Care Section of the American Academy of Optometry.
1. Bielory L. Ocular allergy. Mt Sinai J Med. 2011 Sep-Oct;78(5):740-58.
2. Weismann BA. Giant papillary conjunctivitis. Medscape. 2011 May 26. Available at:
http://emedicine.medscape.com/article/1191641_overview. Accessed January 3, 2013.
3. Friedlaender MH. Ocular allergy. Curr Opin Allergy Clin Immunol. 2011 Oct;11(5):477-82.
4. Elhers WH, Donshik PC. Giant papillary conjunctivitis. Curr Opin Allergy Clin Immunol. 2008 Oct;8(5):445-9.
5. Donshik PC, Porazinski AD. Giant papillary conjunctivitis in frequent-replacement contact lenses wearers: a retrospective study. Am J Ophthalmol. 2000 May; 129(5):703.
6. Ballow M, Donshik PC, Rapacz P, et al. Immunological responses in monkeys to lenses from patients with contact lens-induced giant papillary conjunctivitis. CLAO J. 1989 Jan-Mar;15(1):64-70.
7. Donshik PC, Ehlers WH, Ballow M. Giant papillary conjunctivitis. Immunol Allergy Clin North Am. 2008 Feb;28(1):83-103.
8. Howes JF. Loteprednol etabonate: a review of ophthalmic clinical studies. Pharmazie. 2000 Mar;55(3):178-83.
9. Ohno S, Ando M. A ten-week safety and efficacy evaluation of olopatadine 0.2% instilled twice daily in patients with allergic conjunctivitis in Japan. Nihon Ganka Gakkai Zasshi. 2012 Sep;116(9):869-79.
10. Raizman M. What is new and effective for treating ocular allergies. Eyecare Educators. Available at:
http://www.eyecareeducators.com/site/what_is_new_and_effective_for_treating_ocular_allergies.htm. Accessed January 3, 2013.
11. Khurana S, Sharma N, Agarwal T, et al. Comparison of olopatadine and fluorometholone in contact lens-induced papillary conjunctivitis. Eye Contact Lens. 2010 Jul;36(4):201-4.
12. Kruger CJ, Ehlers WH, Luistro AE, et al. Treatment of giant papillary conjunctivitis with cromolyn sodium. CLAO J. 1992 Jan;18(1):46-8.
13. PorazinskiAD, Donshik PC. Giant papillary conjunctivitis in frequent replacement contact lens wearers: a retrospective study. CLAO J. 1999 Jul;25(3):142-7.


